Should chronic hepatitis B in the indeterminate phase be treated?
-
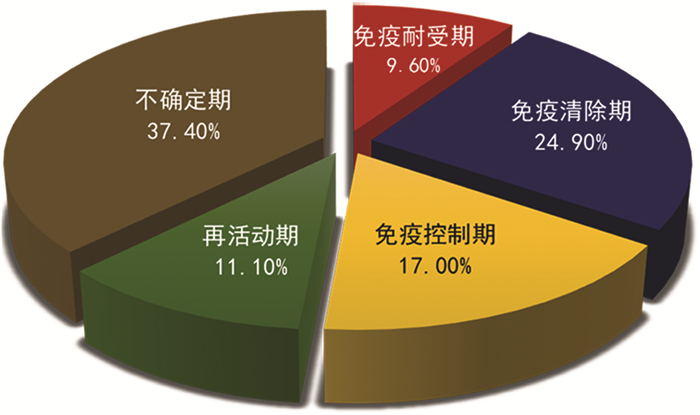
摘要: 探讨了慢性乙型肝炎不确定期的定义,总结了慢性乙型肝炎不确定期在HBV感染者中的占比、发生肝细胞癌风险及治疗建议。Abstract: This article discusses the definition of the indeterminate phase of chronic hepatitis B and summarizes the proportion of patients in the indeterminate phase of chronic hepatitis B among patients with chronic HBV infection, as well as their risk of hepatocellular carcinoma and related treatment recommendations.
-
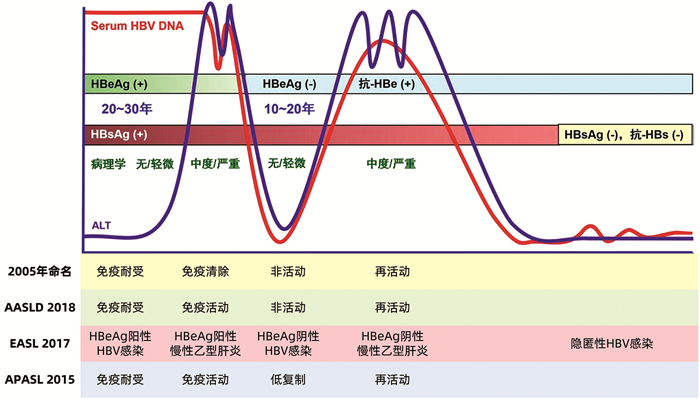
表 1 AASLD、EASL、APASL指南对慢性HBV感染分期的定义
指南 免疫耐受期 免疫活动期(HBeAg阳性) 非活动期 再活动期(HBeAg阴性) AASLD(2018)[1] ALT<ULN或轻度升高,HBV DNA>106 IU/ml,轻度炎症、无纤维化 ALT>2×ULN,HBV DNA≥2×105 IU/ml,≥中度炎症、有或无肝纤维化 ALT<ULN,HBV DNA<2000 IU/ml,肝活检无显著炎症 ALT≥2×ULN,HBV DNA≥2000 IU/ml,中/重度炎症、有或无肝纤维化 EASL(2017)[2] ALT<ULN,HBV DNA高水平,轻度炎症、无纤维化 ALT>ULN,HBV DNA高水平,≥中度炎症、有或无肝纤维化 ALT<ULN,HBV DNA<2000 IU/ml,肝活检无显著炎症 ALT>ULN,HBV DNA中高水平,有炎症和肝纤维化 APASL(2015)[3] ALT<ULN,HBV DNA>2×107 IU/ml,轻微组织学改变 ALT>ULN,HBV DNA持续或间歇高水平,≥中度炎症、有或无肝纤维化 ALT<ULN,HBV DNA低水平,肝活检无显著炎症 ALT>ULN,HBV DNA可测到,持续坏死炎症 表 2 2018 AASLD指导的分期及不符合4期诊断标准的患者
免疫耐受期(HBeAg阳性) 免疫活动期(HBeAg阳性) 非活动期(HBeAg阴性) 再活动期(HBeAg阴性) ALT<ULN或轻度升高,HBV DNA>106 IU/ml,轻度炎症、无纤维化 ALT>2×ULN,HBV DNA≥2×105 IU/ml,≥中度炎症、有或无肝纤维化 ALT<ULN,HBV DNA<2000IU/ml,肝活检无显著炎症 ALT≥2×ULN,HBV DNA≥2000 IU/ml,中/重度炎症、有或无肝纤维化 不符合免疫耐受期,也不符合其他3个期诊断标准患者:(1)其他均符合免疫耐受期标准,但HBV DNA<106 IU/ml(2)其他均符合免疫耐受期标准,但肝脏有显著炎症和纤维化 不属于免疫活动期,也不符合其他3个期诊断标准患者:(1)其他均符合免疫活动期标准,但HBV DNA<2×105 IU/ml(2)其他均符合免疫活动期标准,但ALT 1~2×ULN 不属于非活动期,也不符合其他3个期诊断标准患者:(1)其他均符合非活动期标准,但HBV DNA>2000 IU/ml(2)其他均符合非活动期标准,但有显著炎症和纤维化 不属于再活动期,也不符合其他3个期诊断标准患者:(1)其他均符合再活动期标准,但HBV DNA<2000 IU/ml(2)其他均符合再活动期标准,但ALT 1~2×ULN 表 3 我国慢性HBV感染自然病程分期
检测项目 免疫耐受期(慢性HBV携带状态) 免疫清除期(HBeAg阳性慢性乙型肝炎) 免疫控制期(非活动HBsAg携带状态) 再活动期(HBeAg阴性慢性乙型肝炎) HBV血清标志物 HBsAg(IU/ml) >1×104 + <1×103 + 抗-HBs - - - - HBeAg + + - +/- 抗-HBe - - + +/- 抗-HBc + + + + HBV DNA(IU/ml) >2×107 >2×104 <2×103 >2×103 ALT 正常 持续或反复升高 正常 持续或反复升高 肝脏病理学 无明显炎症坏死和纤维化 有明显炎症坏死和/或纤维化 无或仅有轻度炎症,可有不同程度纤维化 有明显炎症坏死,和/或纤维化 -
[1] TERRAULT NA, LOK A, MCMAHON BJ, et al. Update on prevention, diagnosis, and treatment of chronic hepatitis B: AASLD 2018 hepatitis B guidance[J]. Hepatology, 2018, 67(4): 1560-1599. DOI: 10.1002/hep.29800. [2] European Association for the Study of the Liver. EASL 2017 clinical practice guidelines on the management of hepatitis B virus infection[J]. J Hepatol, 2017, 67(2): 370-398. DOI: 10.1016/j.jhep.2017.03.021. [3] SARIN SK, KUMAR M, LAU GK, et al. Asian-Pacific clinical practice guidelines on the management of hepatitis B: A 2015 update[J]. Hepatol Int, 2016, 10(1): 1-98. DOI: 10.1007/s12072-015-9675-4. [4] HUANG DQ, LI X, LE MH, et al. Natural history and hepatocellular carcinoma risk in untreated chronic hepatitis B patients with indeterminate phase[J]. Clin Gastroenterol Hepatol, 2021. DOI: 10.1016/j.cgh.2021.01.019.[Onlineaheadofprint] [5] SPRADLING PR, XING J, RUPP LB, et al. Distribution of disease phase, treatment prescription and severe liver disease among 1598 patients with chronic hepatitis B in the Chronic Hepatitis Cohort Study, 2006-2013[J]. Aliment Pharmacol Ther, 2016, 44(10): 1080-1089. DOI: 10.1111/apt.13802. [6] HSU YN, PAN CQ, ABBASI A, et al. Clinical presentation and disease phases of chronic hepatitis B using conventional versus modified ALT criteria in Asian Americans[J]. Dig Dis Sci, 2014, 59(4): 865-871. DOI: 10.1007/s10620-014-3054-1. [7] YAO K, LIU J, WANG J, et al. Distribution and clinical characteristics of patients with chronic hepatitis B virus infection in the grey zone[J]. J Viral Hepat, 2021, 28(7): 1025-1033. DOI: 10.1111/jvh.13511. [8] Chinese Society of Infectious Diseases, Chinese Medical Association, Chinese Society of Hepatology, Chinese Medical Association. Guidelines for the prevention and treatment of chronic hepatitis B(version 2019)[J]. J Clin Hepatol, 2019, 35(12): 2648-2669. DOI: 10.3969/j.issn.1001-5256.2019.12.007.中华医学会感染病学分会, 中华医学会肝病学分会. 慢性乙型肝炎防治指南(2019年版)[J]. 临床肝胆病杂志, 2019, 35(12): 2648-2669. DOI: 10.3969/j.issn.1001-5256.2019.12.007. [9] DUAN M, CHI X, XIAO H, et al. High-normal alanine aminotransferase is an indicator for liver histopathology in HBeAg-negative chronic hepatitis B[J]. Hepatol Int, 2021, 15(2): 318-327. DOI: 10.1007/s12072-021-10153-2. -



 PDF下载 ( 3618 KB)
PDF下载 ( 3618 KB)

 下载:
下载: