经肝动脉化疗栓塞术中应用三氧化二砷载药微球与三氧化二砷碘化油治疗不可切除原发性肝癌的成本效益分析
DOI: 10.3969/j.issn.1001-5256.2021.09.024
A cost-effect analysis of transarterial chemoembolization with CalliSpheres beads loaded with arsenic trioxide versus arsenic trioxide iodized oil emulsion in treatment of unresectable liver cancer
-
摘要:
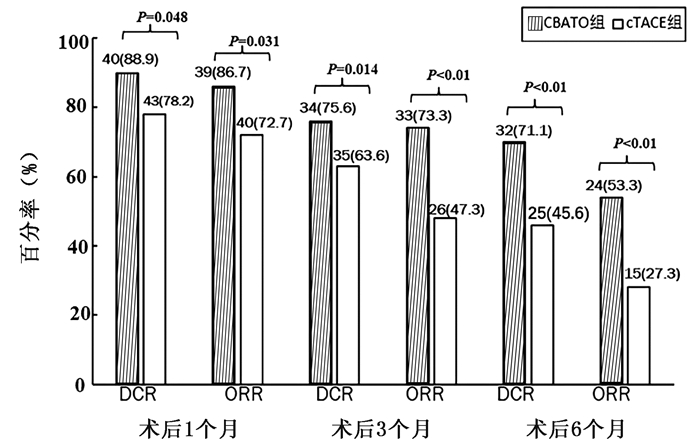
目的 探讨经肝动脉化疗栓塞术(TACE)中应用三氧化二砷(ATO)载药栓塞微球和ATO碘油乳剂栓塞治疗不可切除的肝癌成本效益。 方法 选择2017年5月—2018年12月在郑州大学第一附属医院就诊的100例中晚期原发性肝癌患者作为研究对象,根据治疗方案不同分为CBATO组(n=45)和cTACE组(n=55)。采用无进展生存期(PFS)进行质量调整寿命年(QALY)的疗效评价,采用欧洲五维健康量表(EQ-5D)指数评估患者的生活质量。计量资料2组间比较采用t检验,计数资料2组间比较采用χ2检验,计算2组患者的手术次数、住院时间、治疗成本和增量成本效果比(ICER),并进行成本效益分析。 结果 在PFS内,CBATO组和cTACE组治疗中晚期肝癌的人均住院费用分别为96 446元和91 230.43元。2组间平均手术次数(2.5±0.7 vs 3.4±0.8)和平均住院时间[(5.8±1.2) d vs (7.5±1.8) d]比较,差异均有统计学意义(t值分别为16.911、12.459,P值均<0.01);CBATO组QALY高于cTACE组(0.804>0.512)。与cTACE组相比,CBATO组治疗不可切除肝癌的增量成本效益比为17 861.53元/年。 结论 CBATO组手术花费虽高于cTACE组,但在减少手术次数和住院时间的同时其临床疗效优于cTACE组,术后患者的生活质量高于cTACE组,具有明显的成本效益优势。 Abstract:Objective To investigate the cost-effect of transarterial chemoembolization (TACE) with CalliSpheres beads loaded with arsenic trioxide (ATO) (CBATO) versus ATO iodized oil emulsion (conventional TACE, cTACE) in the treatment of unresectable liver cancer. Methods A total of 100 patients with advanced liver cancer who attended The First Affiliated Hospital of Zhengzhou University from May 2017 to December 2018 were enrolled and divided into CBATO group(n=45) and cTACE group(n=55) according to the treatment regimen. Progression-free survival (PFS) was used to evaluate the efficacy of quality-adjusted life year (QALY), and European Quality of Life-5 Dimensions (EQ-5D) index was used to evaluate quality of life. The t-test was used for comparison of continuous data between groups, and the chi-square test was used for comparison of categorical data between groups; the number of surgeries, length of hospital stay, treatment cost, and incremental cost-effectiveness ratio (ICER) were calculated for the two groups, and then a cost-effect analysis was performed. Results Within the PFS time, the per capita hospital cost was 96 446 yuan in the CBATO group and 91 230.43 yuan in the cTACE group. There were significant differences between the two groups in the mean number of surgeries (2.5±0.7 vs 3.4±0.8, t=16.911, P < 0.01) and mean hospital stay (5.8±1.2 days vs 7.5±1.8 days, t=12.459, P < 0.01). The CBATO group had a significantly higher QALY than the cTACE group (0.804 vs 0.512). Compared with the cTACE group, the CBATO group had an ICER of 17 861.53 yuan/QALY for unresectable liver cancer. Conclusion Although CBATO has a higher surgery cost than cTACE, CBATO has a better clinical effect than cTACE and can reduce the number of surgeries and length of hospital stay, with a better postoperative quality of life than cTACE, suggesting that CBATO has marked cost-effect advantages. -
Key words:
- Liver Neoplasms /
- Chemoembolization, Therapeutic /
- Microspheres /
- Iodized Oil /
- Cost-Benefit Analysis
-
表 1 2组患者的一般资料比较
项目 CBATO组(n=45) cTACE(n=55) 统计值 P值 年龄(岁) 58.94±9.89 55.14±9.03 t=0.478 0.491 性别[例(%)] χ2=0.112 0.738 男 41(91.1) 49(89.1) 女 4(8.9) 6(10.9) 病因学[例(%)] χ2=0.455 0.500 HBV 39(86.7) 50(90.9) 其他 6(13.3) 5(9.1) ECOG[例(%)] χ2=0.026 0.872 0 23(51.1) 29(52.7) 1 22(48.9) 26(47.3) Child分级[例(%)] χ2=2.899 0.089 A 39(86.7) 40(72.7) B 6(13.3) 15(27.3) 肝硬化[例(%)] χ2=1.010 0.315 是 38(84.5) 42(76.4) 否 7(15.5) 13(23.6) BCLC分期[例(%)] χ2=0.004 0.952 B 24(53.3) 29(52.7) C 21(46.7) 26(47.3) 肿瘤大小[例(%)] χ2=0.134 0.714 ≤5 cm 27(60.0) 31(56.4) >5 cm 18(40.0) 24(43.6) 表 2 CBATO组与cTACE组直接医疗费用比较
分类 CBATO组 cTACE组 差异 综合医疗服务费用(元) 床位费 232 376 -144 护理费 169 258 -89 一般医疗服务费 489 1184 -695 诊断费用(元) 实验室诊断费用 929.67 1 489.90 -560.23 影像学诊断费用 2 002.75 2 322.21 -319.46 术中花费(元) 其他术中花费(手术费+器械费) 11 727.04 12 605.94 521.10 栓塞材料费 21 562.43 5 997.89 15 564.54 并发症处理花费(元) 1 466.51 2 598.54 -1 132.03 合计(元) 38 578.4 26 832.48 11 745.92 注:差异为CBATO组的花费减去cTACE组的花费。 表 3 EQ-5D指数表
时间点 CBATO组(n=45) cTACE组(n=55) t值 P值 术前 0.57±0.12 0.60±0.13 1.545 0.219 术后1个月 0.71±0.19 0.67±0.17 6.379 0.014 术后3个月 0.77±0.11 0.70±0.18 28.302 <0.01 术后6个月 0.76±0.11 0.69±0.10 28.212 <0.01 表 4 2组治疗方案成本-效益比较
组别 总成本(元) 效益(%) 成本/效益比 QALY(元/QALY) ICER(元/QALY) CBATO组 96 446.00 71.1 1 356.48 0.804 17 861.53 cTACE组 91 230.43 45.6 2 000.67 0.512 表 5 患者平均住院时间和手术次数对比
组别 手术次数(次) 单次住院天数(d) CBATO组 2.5±0.7 5.8±1.2 cTACE组 3.4±0.8 7.5±1.8 t值 16.911 12.459 P值 <0.01 <0.01 -
[1] LAI CL, RATZIU V, YUEN MF, et al. Viral hepatitis B[J]. Lancet, 2003, 362(9401): 2089-2094. DOI: 10.1016/S0140-6736(03)15108-2. [2] European Association for the Study of the Liver. EASL clinical practice guidelines: Management of hepatocellular carcinoma[J]. J Hepatol, 2018, 69(1): 182-236. DOI: 10.1016/j.jhep.2018.03.019. [3] AKO S, NAKAMURA S, NOUSO K, et al. Transcatheter arterial chemoembolization to reduce size of hepatocellular carcinoma before radiofrequency ablation[J]. Acta Med Okayama, 2018, 72(1): 47-52. DOI: 10.18926/AMO/55662. [4] SONG MJ, CHUN HJ, SONG DS, et al. Comparative study between doxorubicin-eluting beads and conventional transarterial chemoembolization for treatment of hepatocellular carcinoma[J]. J Hepatol, 2012, 57(6): 1244-1250. DOI: 10.1016/j.jhep.2012.07.017. [5] FACCIORUSSO A, DI MASO M, MUSCATIELLO N. Drug-eluting beads versus conventional chemoembolization for the treatment of unresectable hepatocellular carcinoma: A meta-analysis[J]. Dig Liver Dis, 2016, 48(6): 571-577. DOI: 10.1016/j.dld.2016.02.005. [6] PESAPANE F, NEZAMI N, PATELLA F, et al. New concepts in embolotherapy of HCC[J]. Med Oncol, 2017, 34(4): 58. DOI: 10.1007/s12032-017-0917-2. [7] National Health and Family Planning Commission of the People's Republic of China. Diagnosis, management, and treatment of hepatocellular carcinoma (V2017)[J]. J Clin Hepatol, 2017, 33(8): 1419-1431. DOI: 10.3969/j.issn.1001-5256.2017.08.003.中华人民共和国国家卫生和计划生育委员会. 原发性肝癌诊疗规范(2017年版)[J]. 临床肝胆病杂志, 2017, 33(8): 1419-1431. DOI: 10.3969/j.issn.1001-5256.2017.08.003. [8] DUAN XH, JU SG, HAN XW, et al. Arsenic trioxide-eluting Callispheres beads is more effective and equally tolerant compared with arsenic trioxide/lipiodol emulsion in the transcatheter arterial chemoembolization treatment for unresectable hepatocellular carcinoma patients[J]. Eur Rev Med Pharmacol Sci, 2020, 24(3): 1468-1480. DOI: 10.26355/eurrev_202002_20206. [9] FISMAN DN, TUITE AR. Estimation of the health impact and cost-effectiveness of influenza vaccination with enhanced effectiveness in Canada[J]. PLoS One, 2011, 6(11): e27420. DOI: 10.1371/journal.pone.0027420. [10] NEUMANN PJ, SANDERS GD. Cost-effectiveness analysis 2.0[J]. N Engl J Med, 2017, 376(3): 203-205. DOI: 10.1056/NEJMp1612619. [11] YIN WL, LIAN J, XIAO SX, et al. Clinical effect of drug-eluting beads and conventional transcatheter arterial chemoembolization in treatment of unresectable liver cancer: A meta-analysis[J]. J Clin Hepatol, 2019, 35(6): 1270-1275. DOI: 10.3969/j.issn.1001-5256.2019.06.018.尹伟利, 连佳, 肖时湘, 等. 载药微球与传统碘化油经肝动脉化疗栓塞术治疗不可切除肝癌效果比较的Meta分析[J]. 临床肝胆病杂志, 2019, 35(6): 1270-1275. DOI: 10.3969/j.issn.1001-5256.2019.06.018. [12] FORNER A, GILABERT M, BRUIX J, et al. Treatment of intermediate-stage hepatocellular carcinoma[J]. Nat Rev Clin Oncol, 2014, 11(9): 525-535. DOI: 10.1038/nrclinonc.2014.122. [13] MELCHIORRE F, PATELLA F, PESCATORI L, et al. DEB-TACE: A standard review[J]. Future Oncol, 2018, 14(28): 2969-2984. DOI: 10.2217/fon-2018-0136. [14] MALAGARI K, POMONI M, MOSCHOURIS H, et al. Chemoembolization of hepatocellular carcinoma with HepaSphere 30-60 μm. Safety and efficacy study[J]. Cardiovasc Intervent Radiol, 2014, 37(1): 165-175. DOI: 10.1007/s00270-013-0777-x. [15] CHEN MS, HU ZL. Research progress of hepatic arterial infusion chemotherapy in the conversion therapy of hepatocellular carcinoma[J]. Chin J Dig Surg, 2021, 20(2): 171-177. DOI: 10.3760/cma.j.cn115610-20201230-00813.陈敏山, 胡自力. 肝动脉灌注化疗在肝癌转化治疗中的研究进展[J]. 中华消化外科杂志, 2021, 20(2): 171-177. DOI: 10.3760/cma.j.cn115610-20201230-00813. [16] TRAN BX, OHINMAA A, NGUYEN LT. Quality of life profile and psychometric properties of the EQ-5D-5L in HIV/AIDS patients[J]. Health Qual Life Outcomes, 2012, 10: 132. DOI: 10.1186/1477-7525-10-132. [17] XING M, WEBBER G, PRAJAPATI HJ, et al. Preservation of quality of life with doxorubicin drug-eluting bead transarterial chemoembolization for unresectable hepatocellular carcinoma: Longitudinal prospective study[J]. J Gastroenterol Hepatol, 2015, 30(7): 1167-1174. DOI: 10.1111/jgh.12920. [18] LIU YS, OU MC, TSAI YS, et al. Transarterial chemoembolization using gelatin sponges or microspheres plus lipiodol-doxorubicin versus doxorubicin-loaded beads for the treatment of hepatocellular carcinoma[J]. Korean J Radiol, 2015, 16(1): 125-132. DOI: 10.3348/kjr.2015.16.1.125. [19] ZHU D, YUAN D, WANG Z, et al. Efficacy of drug-eluting bead transarterial chemoembolization (DEB-TACE) combined with radiofrequency ablation versus DEB-TACE alone in Chinese hepatocellular carcinoma patients[J]. Medicine (Baltimore), 2019, 98(26): e15682. DOI: 10.1097/MD.0000000000015682. [20] KIMURA H, WEISZ A, KURASHIMA Y, et al. Hypoxia response element of the human vascular endothelial growth factor gene mediates transcriptional regulation by nitric oxide: Control of hypoxia-inducible factor-1 activity by nitric oxide[J]. Blood, 2000, 95(1): 189-197. -



 PDF下载 ( 2198 KB)
PDF下载 ( 2198 KB)

 下载:
下载:


