妊娠女性药物相关肝损伤风险的识别及分析
DOI: 10.3969/j.issn.1001-5256.2022.08.021
Identification and analysis of the risk of liver-related adverse drug reaction in pregnant women
-
摘要:
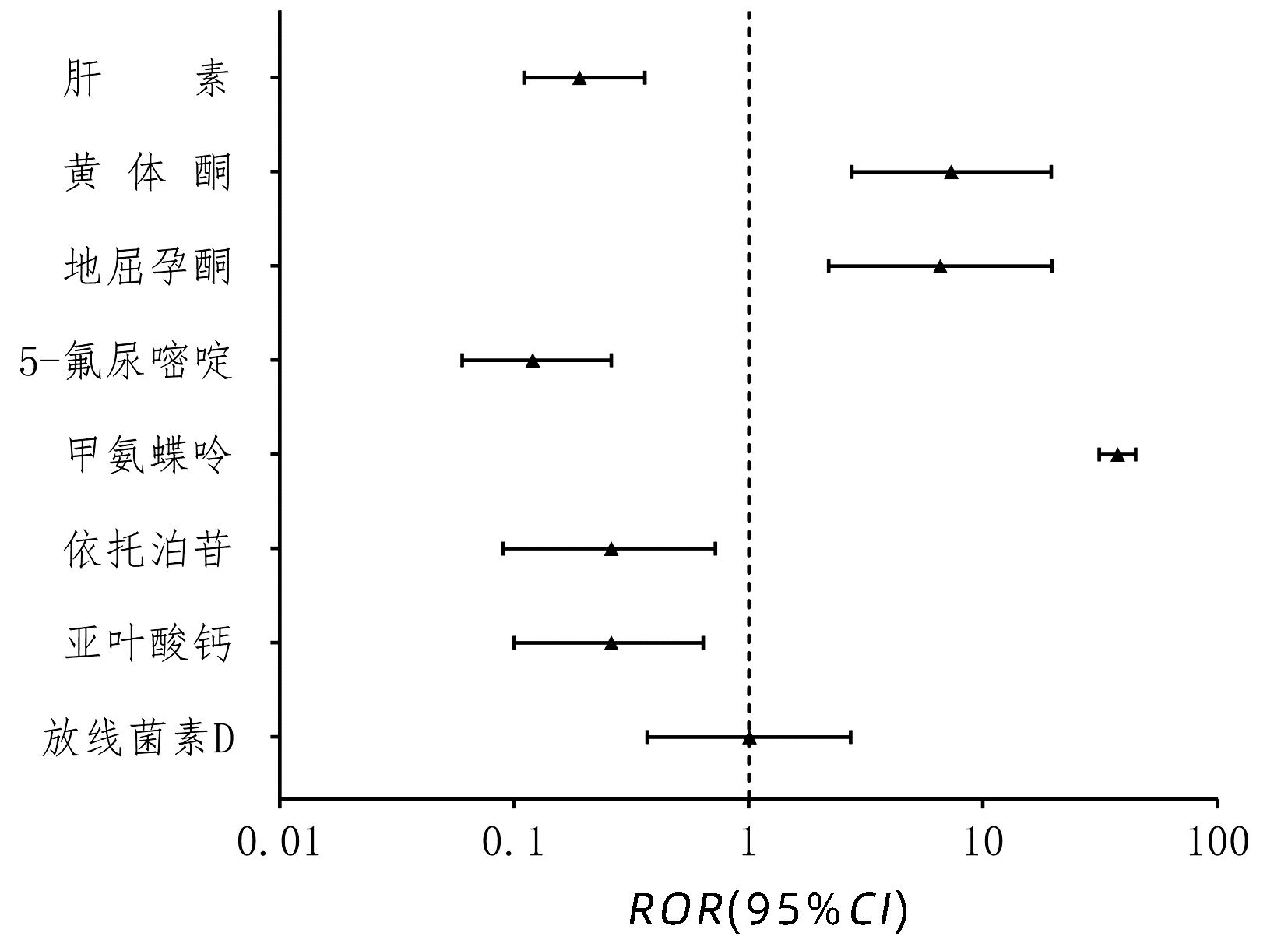
目的 通过对妊娠女性药物相关肝损伤不良反应(ADR)特征识别与分析,警示其潜在用药风险。 方法 收集中草药肝损伤云系统(hilicloud.net)中2012年1月1日—2016年12月31日妊娠女性药物相关肝损伤ADR报告进行回顾性研究,分析其主要临床特征及用药规律,并采用报告比值比(ROR)对相关药物进行相对风险分析。计量资料组间比较采用t检验。 结果 妊娠女性药物相关肝损伤报告的高频药物有甲氨蝶呤、米非司酮、利托君等,主要用于终止异位妊娠及葡萄胎等疾病的治疗。药物相关肝损伤相对风险分析发现,妊娠女性使用甲氨蝶呤(ROR=37.52,95%CI:31.35~44.89)、黄体酮(ROR=7.33,95%CI:2.75~19.59)、地屈孕酮(ROR=6.58,95%CI:2.20~19.69)与肝损伤风险相关性较强,其中妊娠女性(ROR=37.52,95%CI:31.35~44.89)使用甲氨蝶呤比非妊娠女性(ROR=1.71,95%CI:1.47~4.36)使用发生肝损伤的风险相关性强。 结论 应重视甲氨蝶呤、黄体酮等药物在妊娠女性人群中发生肝损伤的潜在风险,避免严重不良反应的发生。 -
关键词:
- 妊娠 /
- 化学性与药物性肝损伤 /
- 药物相关性副作用和不良反应
Abstract:Objective To investigate the potential medication risk by identifying and analyzing the features of liver-related adverse drug reaction (ADR) in pregnant women. Methods A retrospective study was performed for the reports on liver-related ADR in pregnant women from January 1, 2012 to December 31, 2016 in HILI Cloud (hilicloud.net). Main clinical features and medication rules were analyzed, and reporting odds ratio (ROR) was used to analyze the relative risk of related drugs. Results Methotrexate, mifepristone, and ritodrine were the high-frequency drugs reported for liver-related ADR in pregnant women and were mainly used for termination of ectopic pregnancy and treatment of hydatidiform mole. The relative risk analysis of liver-related ADR showed that in pregnant women, the use of methotrexate (ROR=37.52, 95% confidence interval [CI]=31.35-44.89), progesterone (ROR=7.33, 95%CI: 2.75-19.59), and dydrogesterone (ROR=6.58, 95%CI: 2.20-19.69) was strongly associated with the risk of liver injury, and the association of methotrexate with the risk of liver injury in pregnant women was significantly stronger than that in non-pregnant women (ROR=1.71, 95%CI: 1.47-4.36). Conclusion The potential risk of liver injury should be taken seriously in pregnant women using the drugs such as methotrexate and progesterone, so as to avoid serious adverse reactions. -
表 1 药物相关肝损伤ADR报告频数排名前十的药物
Table 1. Reporting frequency of liver-related ADR for Top 10
药物 报告数(例) 占比(%) 药物 报告数(例) 占比(%) 甲氨蝶呤 871 78.33 地屈孕酮 9 0.81 米非司酮 135 12.14 5-氟尿嘧啶 7 0.63 利托君 13 1.17 亚叶酸钙 5 0.45 黄体酮 12 1.08 放线菌素D 5 0.45 肝素 11 0.99 依托泊苷 4 0.36 -
[1] World Health Organization. World patient safety day 2021[EB/OL]. https://www.who.int/newsroom/events/detail/2021/09/17/defaultcalendar/world patient safety day 2021. [2] ITO S. Mother and child: medication use in pregnancy and lactation[J]. Clin Pharmacol Ther, 2016, 100(1): 8-11. DOI: 10.1002/cpt.383. [3] LI L. Effects of puerarin with insulin aspart on biochemical indexes and artery blood flow parameters related to the fetus of pregnant women with Gestational diabetes[J]. World Chi Med, 2018, 13(5): 1134-1138. DOI: 10.3969/j.issn.1673-7202.2018.05.021.李玲. 葛根素联合门冬胰岛素对妊娠期糖尿病孕妇生化指标及胎儿相关动脉血流参数的影响[J]. 世界中医药, 2018, 13(5): 1134-1138. DOI: 10.3969/j.issn.1673-7202.2018.05.021. [4] World Health Organization, U. M. C. The use of the WHO-UMC system for standardised case causality assessment[EB/OL]. https://www.who-umc.org/media/164200/who-umc-causality-assessment_new-logo.pdf. [5] XIAO XH, TANG JM, MAO YM, et al. Guiding principles of clinical evaluation technology of traditional Chinese medicine induced liver injury[J]. Acta Pharm Sin, 2018, 53(11): 1931-1942. https://www.cnki.com.cn/Article/CJFDTOTAL-YXXB201811021.htm肖小河, 唐健元, 茅益民, 等. 中药药源性肝损伤临床评价技术指导原则[J]. 药学学报, 2018, 53(11): 1931-1942. https://www.cnki.com.cn/Article/CJFDTOTAL-YXXB201811021.htm [6] HOU Y, YE X, WU G, et al. A comparison of disproportionality analysis methods in national adverse drug reaction databases of China[J]. Expert Opin Drug Saf, 2014, 13(7): 853-857. DOI: 10.1517/14740338.2014.915938. [7] van PUIJENBROEK EP, BATE A, LEUFKENS HG, et al. A comparison of measures of disproportionality for signal detection in spontaneous reporting systems for adverse drug reactions[J]. Pharmacoepidemiol Drug Saf, 2002, 11(1): 3-10. DOI: 10.1002/pds.668. [8] MENG L, TANG XW, JI HH, et al. Mining and evaluation of statin-associated adverse events signals: data mining of the public version of the OpenFDA adverse event reporting system[J]. Chin J New Drugs, 2019, 28(2): 244-248. https://www.cnki.com.cn/Article/CJFDTOTAL-ZXYZ201902019.htm蒙龙, 唐学文, 季欢欢, 等. 他汀类药物相关不良反应的信号挖掘与评价——基于美国OpenFDA公共数据开放项目的数据挖掘研究[J]. 中国新药杂志, 2019, 28(2): 244-248. https://www.cnki.com.cn/Article/CJFDTOTAL-ZXYZ201902019.htm [9] REN JT, WANG SF, HOU YF, et al. Common signal detection methods of adverse drug reaction[J]. J Pharm, 2011, 8(5): 294-298. DOI: 10.3969/j.issn.1672-8629.2011.05.013.任经天, 王胜锋, 侯永芳, 等. 常用药品不良反应信号检测方法介绍[J]. 中国药物警戒, 2011, 8(5): 294-298. DOI: 10.3969/j.issn.1672-8629.2011.05.013. [10] ZHANG JY, BAI YX, HAN S, et al. Application of data mining algorithm to detect adverse drug reaction signal[J]. ADRJ, 2016, 18(6): 412-416. DOI: 10.3760/cma.j.issn.1008-5734.2016.06.003.张婧媛, 白羽霞, 韩晟, 等. 数据挖掘方法检测药品不良反应信号的应用研究[J]. 药物不良反应杂志, 2016, 18(6): 412-416. DOI: 10.3760/cma.j.issn.1008-5734.2016.06.003. [11] LI SJ, CHEN L, WU S, et al. Analysis and application of measures of dis-proportionality in methotrexate adverse reaction signal mining[J]. Anti-tumor Pharm, 2018, 8(2): 274-278, 287. DOI: 10.3969/j.issn.2095-1264.2018.02.34.李双娇, 陈力, 吴珊, 等. 比值失衡测量法在甲氨蝶呤不良反应信号挖掘中的应用与分析[J]. 肿瘤药学, 2018, 8(2): 274-278, 287. DOI: 10.3969/j.issn.2095-1264.2018.02.34. [12] Ministry of Health, The People's Republic of China. Measures for the administration of adverse drug reaction reporting and monitoring (Order No. 81)[EB/OL]. http://www.gov.cn/zhengce/2011-05/24/content_2603398.htm.中华人民共和国卫生部. 《药品不良反应报告和监测管理办法》(第81号令)[EB/OL]. http://www.gov.cn/zhengce/2011-05/24/content_2603398.htm. [13] SONG SQ, ZHANG GN. Combination chemotherapy of blemycin etoposide cisplatin or carboplatin for the treatment of malignant gestational trophoblastic tumor[J]. Chin J Clin Obstet Gynecol, 2006, (4): 248-250. DOI: 10.3969/j.issn.1672-1861.2006.04.003.宋水勤, 张国楠. BEP方案治疗恶性滋养细胞肿瘤38例临床分析[J]. 中国妇产科临床杂志, 2006, (4): 248-250. DOI: 10.3969/j.issn.1672-1861.2006.04.003. [14] YUAN SW, CEN Y. Diagnosis, treatment and research progress of gestational trophoblastic tumor[J/CD]. J World Latest Med Inf, 2019, 19(48): 324-325. DOI: 10.19613/j.cnki.1671-3141.2019.48.223.贠舒雯, 岑尧. 妊娠滋养细胞肿瘤的诊治及研究进展[J/CD]. 世界最新医学信息文摘, 2019, 19(48): 324-325. DOI: 10.19613/j.cnki.1671-3141.2019.48.223. [15] Gynecological oncology Committee of China Anti Cancer Association. Guidelines for the diagnosis and treatment of gestational trophoblastic diseases (2021 Edition)[J]. China Oncol, 2021, 31(6): 520-532. DOI: 10.19401/j.cnki.1007-3639.2021.06.10.中国抗癌协会妇科肿瘤专业委员会. 妊娠滋养细胞疾病诊断与治疗指南(2021年版)[J]. 中国癌症杂志, 2021, 31(6): 520-532. DOI: 10.19401/j.cnki.1007-3639.2021.06.10. [16] WANG LJ, LIN HX, LIN ZQ. Interpretation of 2021 NCCN clinical practice guide for gestational trophoblastic tumor (2nd Edition)[J]. Chin J Pract Gynecol Obstetr, 2021, 37(5): 564-569. DOI: 10.19538/j.fk2021050115.王丽娟, 林海雪, 林仲秋. 《2021 NCCN妊娠滋养细胞肿瘤临床实践指南(第2版)》解读[J]. 中国实用妇科与产科杂志, 2021, 37(5): 564-569. DOI: 10.19538/j.fk2021050115. [17] LIANG Y, MENG Z, CANG HQ, et al. Acute drug-induced liver injury caused by vancomycin hydrochloride for injection: one case report[J]. J Pharm, 2021, 18(8): 789-792. DOI: 10.19803/j.1672-8629.2021.08.20.梁瑜, 孟真, 仓怀芹, 等. 注射用盐酸万古霉素致急性药物性肝损伤1例分析[J]. 中国药物警戒, 2021, 18(8): 789-792. DOI: 10.19803/j.1672-8629.2021.08.20. [18] WANG XQ, HE YB, QIAN ZY, et al. Molecular mechanism of Nifedipine inducing liver injury in children[J]. J Appl Clin Pediatr, 2018, 33(6): 465-469. DOI: 10.3760/cma.j.issn.2095-428X.2018.06.017.王新启, 贺轶博, 钱致远, 等. 硝苯地平诱发儿童肝损伤的分子机制[J]. 中华实用儿科临床杂志, 2018, 33(6): 465-469. DOI: 10.3760/cma.j.issn.2095-428X.2018.06.017. [19] ZHANG YF, YE XJ, CHEN M, et al. A case of acute liver injury caused by ceftriaxone/tazobactam[J]. Chin J Pharmacoepidemiol, 2021, 30(6): 421-422. https://www.cnki.com.cn/Article/CJFDTOTAL-YWLX202106013.htm张艳芳, 叶旭军, 陈敏, 等. 头孢曲松/他唑巴坦致急性肝损伤1例[J]. 药物流行病学杂志, 2021, 30(6): 421-422. https://www.cnki.com.cn/Article/CJFDTOTAL-YWLX202106013.htm [20] HUANG DL, DAI W. A case of acute drug-induced liver injury caused by norethindrone[J]. J Clin Hepatol, 2018, 34(6): 1294-1295. DOI: 10.3969/j.issn.1001-5256.2018.06.033.黄德良, 戴炜. 炔诺酮致急性药物性肝损伤1例报告[J]. 临床肝胆病杂志, 2018, 34(6): 1294-1295. DOI: 10.3969/j.issn.1001-5256.2018.06.033. [21] WANG Y, YE ZK, CUI XL, et al. Pharmaceutical care of a patient with liver injury induced by omeprazole or octreotide[J]. Chin J Clin Pharmacol, 2017, 33(19): 1959-1960. DOI: 10.13699/j.cnki.1001-6821.2017.19.031.王颖, 叶志康, 崔向丽, 等. 奥美拉唑或奥曲肽致肝损伤1例患者的药学监护[J]. 中国临床药理学杂志, 2017, 33(19): 1959-1960. DOI: 10.13699/j.cnki.1001-6821.2017.19.031. [22] KATSUHISA I, HIROAKI Y. Molecular basis for pharmacokinetics and pharmacodynamics of methotrexate in rheumatoid arthritis therapy[J]. Drug Metab Pharmacokinet, 2014, 29(1): 12-19. DOI: 10.2133/dmpk.dmpk-13-rv-119. [23] WANG W, ZHOU H, LIU L. Side effects of methotrexate therapy for rheumatoid arthritis: A systematic review[J]. Eur J Med Chem, 2018, 158: 502-516. DOI: 10.1016/j.ejmech.2018.09.027. [24] BAI ZF, GAO Y, ZUO XB, et al. Progress in research on the pathogenesis of immune regulation and idiosyncratic drug-induced liver injury[J]. Acta Pharm Sin, 2017, 52(7): 1019-1026. DOI: 10.16438/j.0513-4870.2017-0315.柏兆方, 高源, 左晓彬, 等. 免疫调控与特异质型药物性肝损伤发生机制研究进展[J]. 药学学报, 2017, 52(7): 1019-1026. DOI: 10.16438/j.0513-4870.2017-0315. [25] GUO YM, TU C, HE Q, et al. Safety administration strategy for heshouwu (polygoni multiflori radix) based on properties and actions of Chinese medicine[J]. J Tradit Chin Med, 2018, 59(9): 721-724. DOI: 10.13288/j.11-2166/r.2018.09.001.郭玉明, 涂灿, 何琴, 等. 基于药性功能认知的何首乌安全用药对策研究[J]. 中医杂志, 2018, 59(9): 721-724. DOI: 10.13288/j.11-2166/r.2018.09.001. [26] TSAKIRIDIS I, GIOULEKA S, MAMOPOULOS A, et al. Diagnosis and management of ectopic pregnancy: a comparative review of major national guidelines[J]. Obstet Gynecol Surv, 2020, 75(10): 611-623. DOI: 10.1097/OGX.0000000000000832. [27] PO L, THOMAS J, MILLS K, et al. Guideline No. 414: management of pregnancy of unknown location and tubal and nontubal ectopic pregnancies[J]. J Obstet Gynaecol Can, 2021, 43(5): 614-630. e1. DOI: 10.1016/j.jogc.2021.01.002. [28] HU TX, LYU XQ, LI L, et al. Analysis of 4217 cases of adverse drug reactions induced by progesterone oral agents[J]. J Pharm, 2018, 15(12): 728-732. DOI: 10.3969/j.issn.1672-8629.2018.12.005.胡婷霞, 吕小琴, 李岚, 等. 4217例黄体酮口服制剂不良反应报告分析[J]. 中国药物警戒, 2018, 15(12): 728-732. DOI: 10.3969/j.issn.1672-8629.2018.12.005. -



 PDF下载 ( 2634 KB)
PDF下载 ( 2634 KB)


 下载:
下载: