生长分化因子11(GDF11)调节非酒精性脂肪性肝病的机制研究
DOI: 10.3969/j.issn.1001-5256.2023.09.011
利益冲突声明:本文不存在任何利益冲突。
作者贡献声明:张涛负责课题设计,实验实施,收集数据,资料分析,撰写论文;殷雪翠参与采集数据,分析数据;任飞飞、董琳、张佳参与统计分析;卢高峰参与课题设计,审核论文并对论文负责。
Mechanism of growth differentiation factor 11 regulating nonalcoholic fatty liver disease
-
摘要:
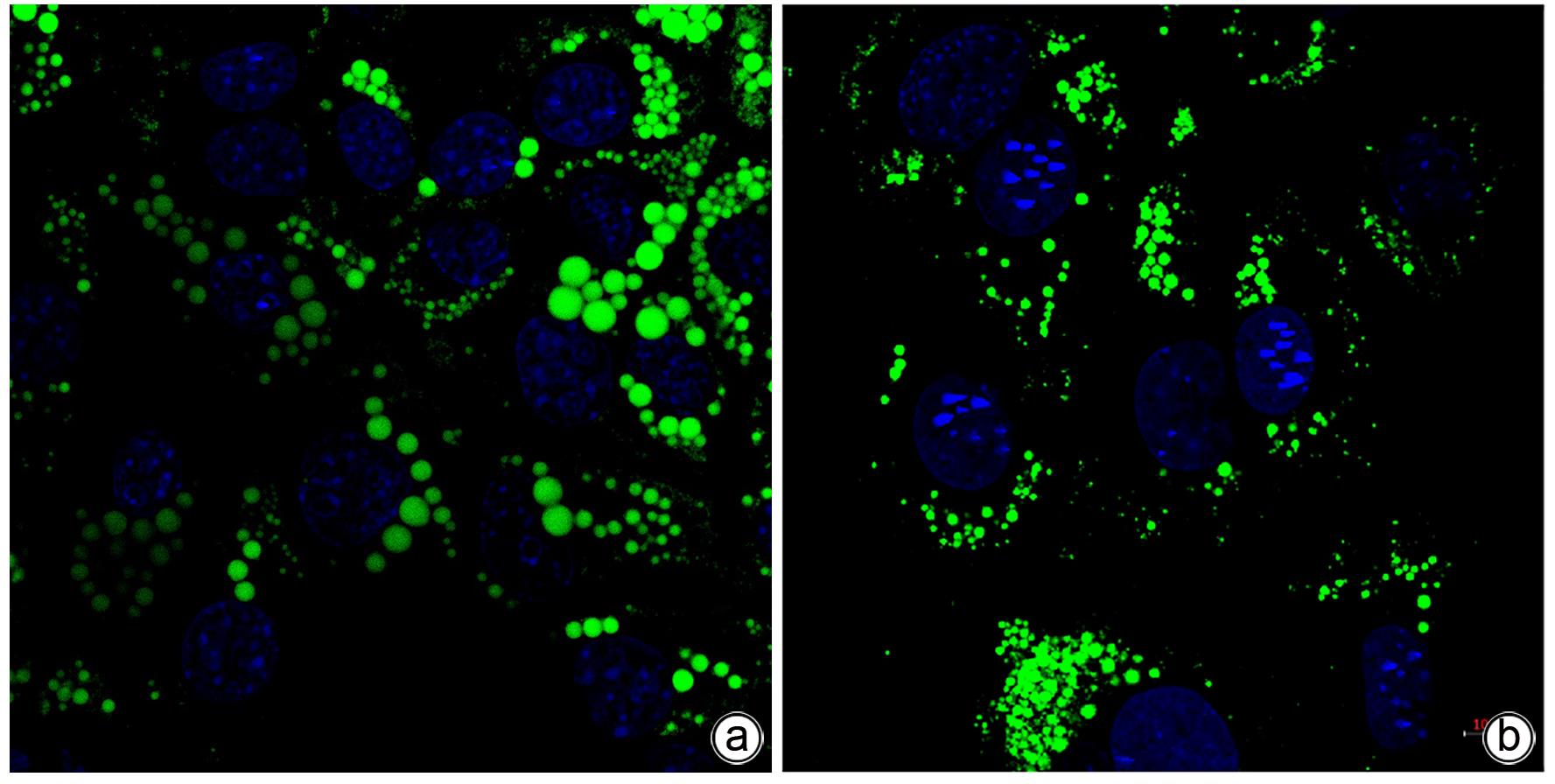
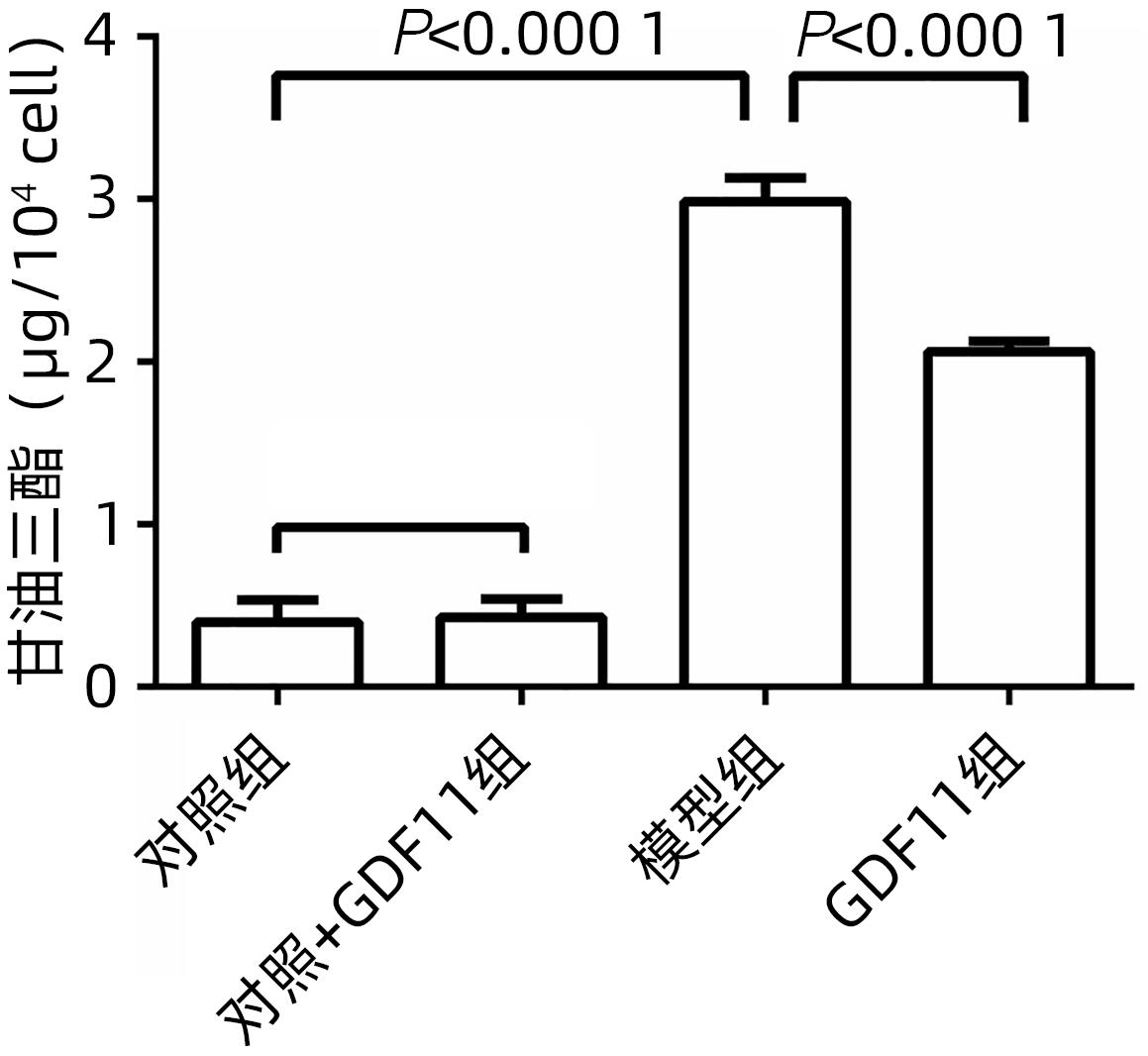
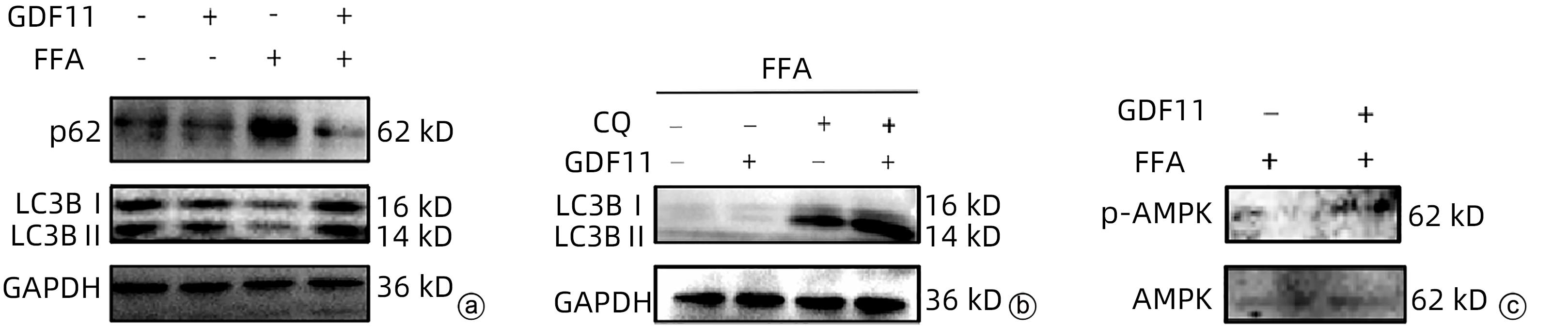
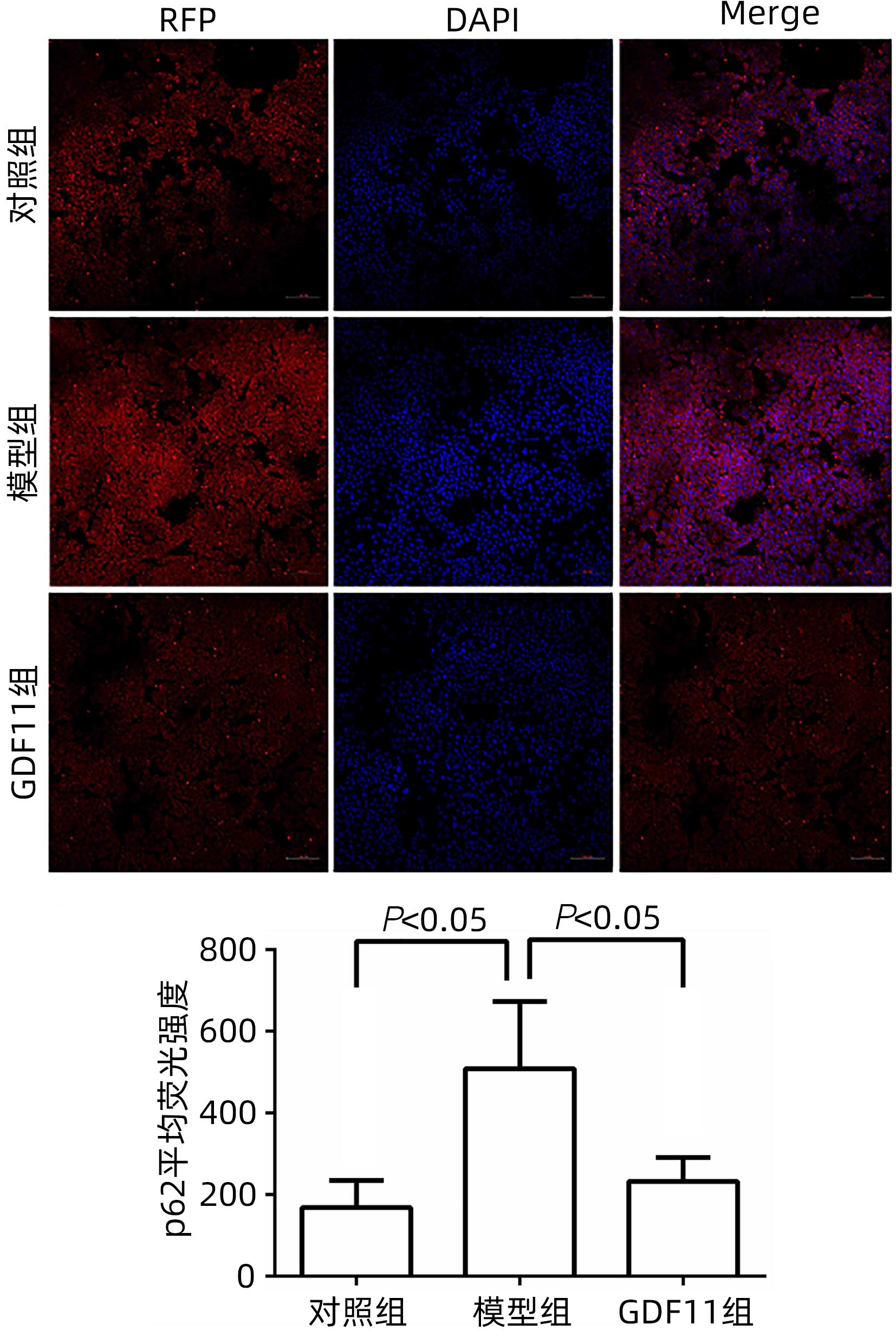
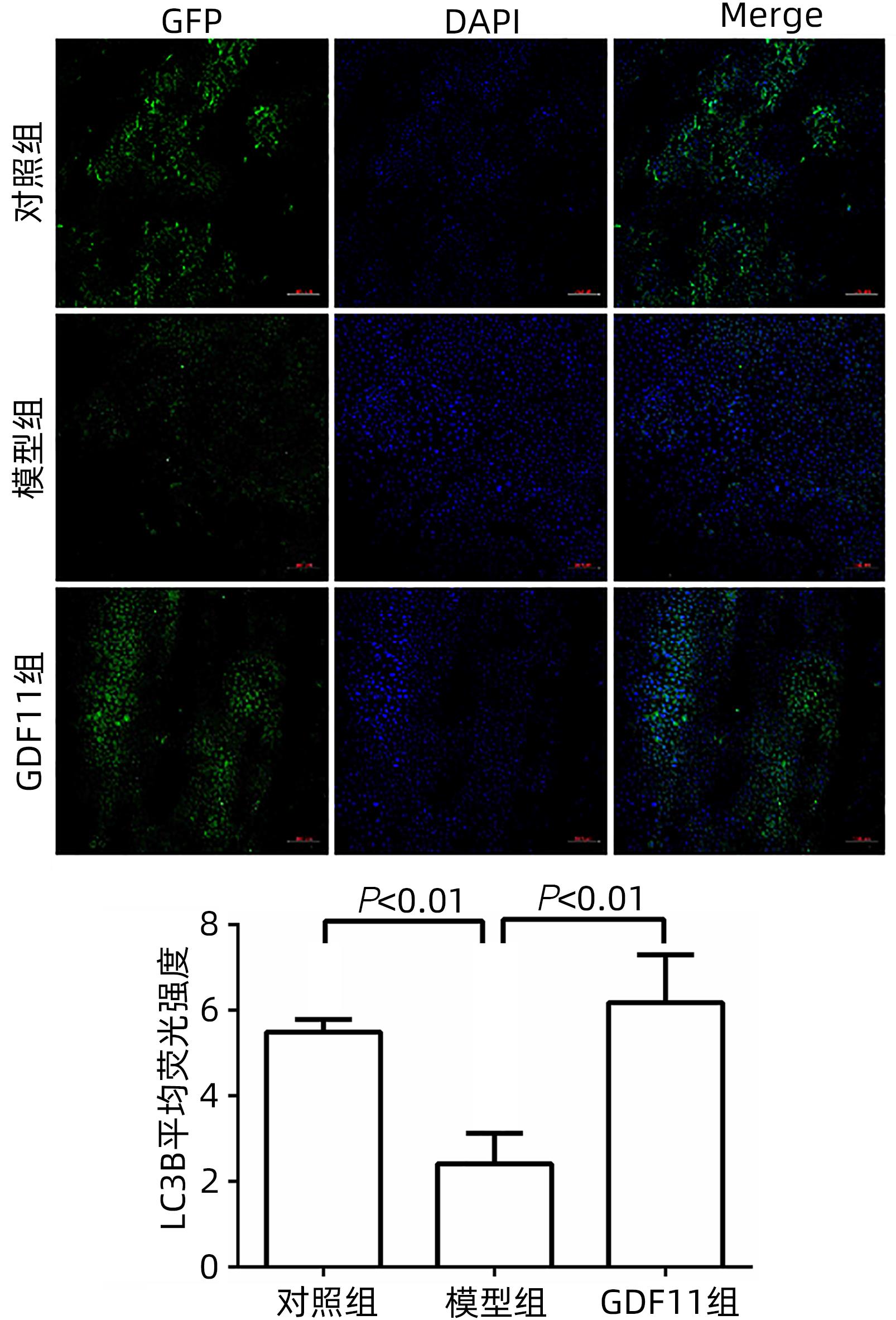
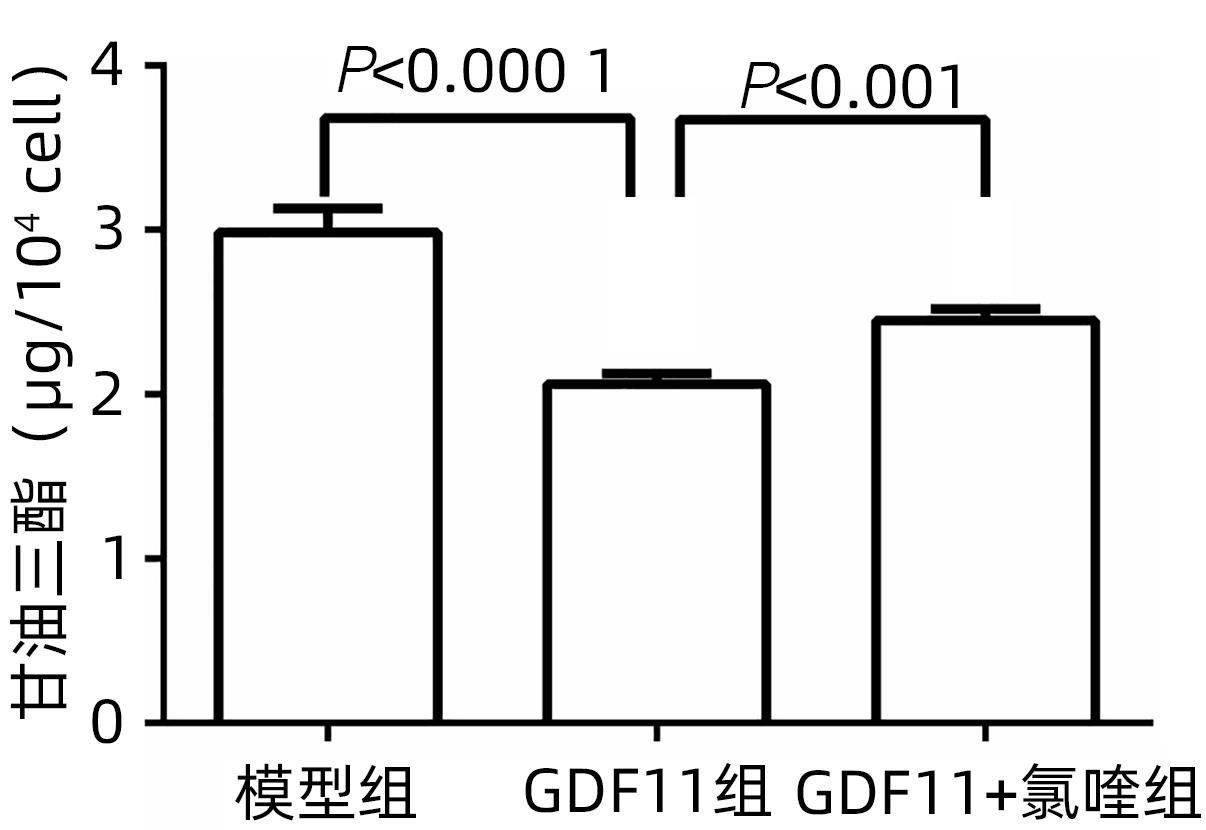
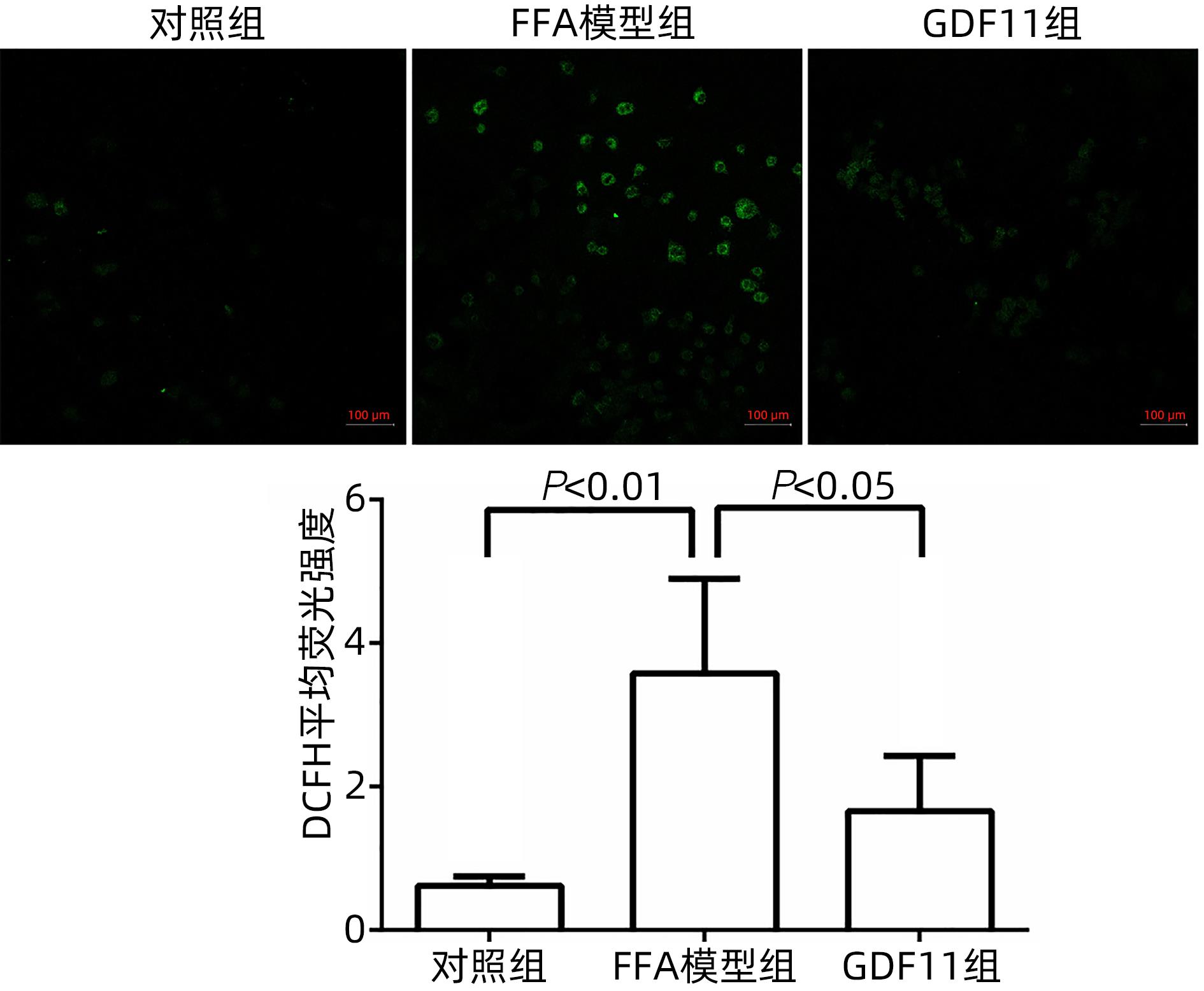
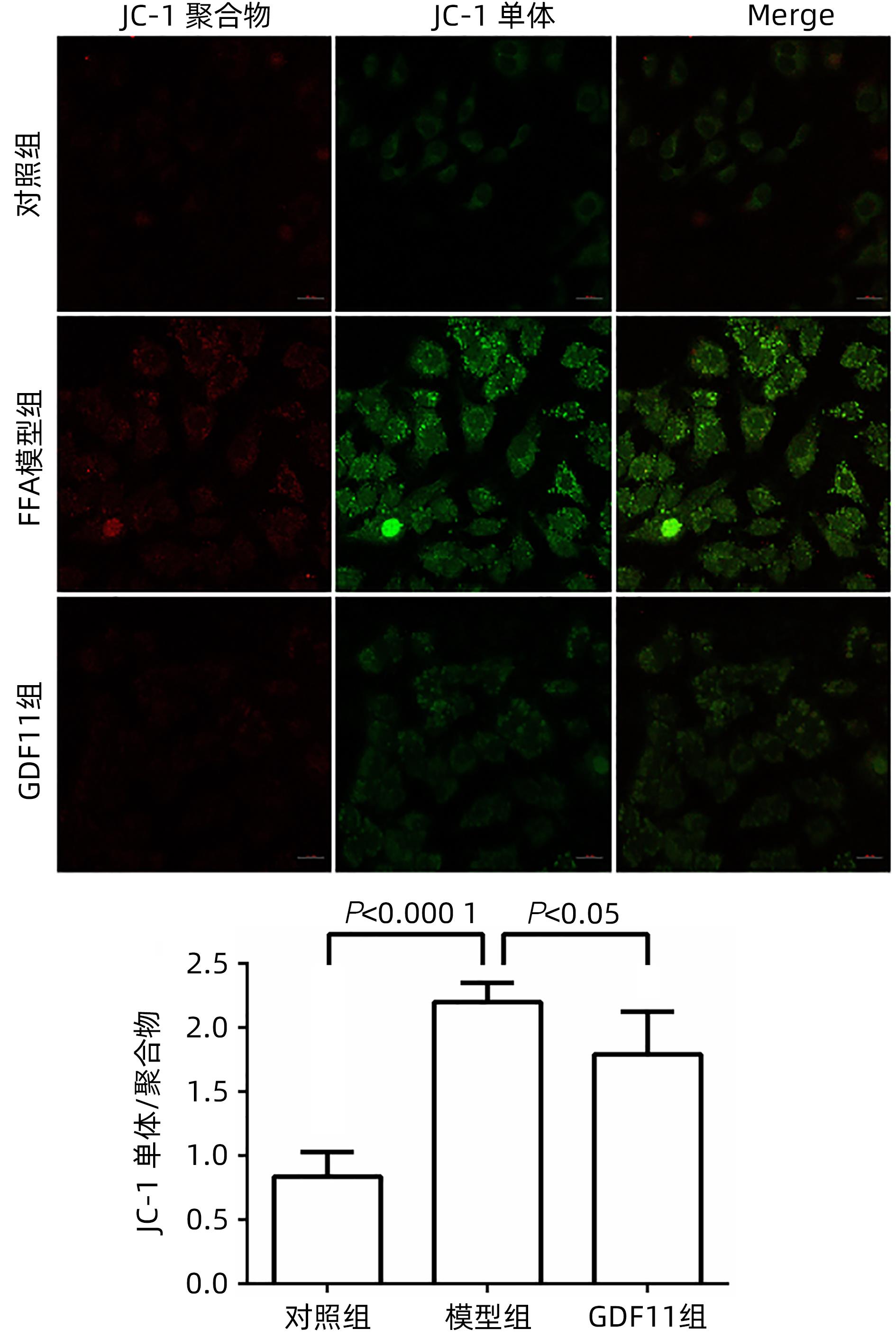
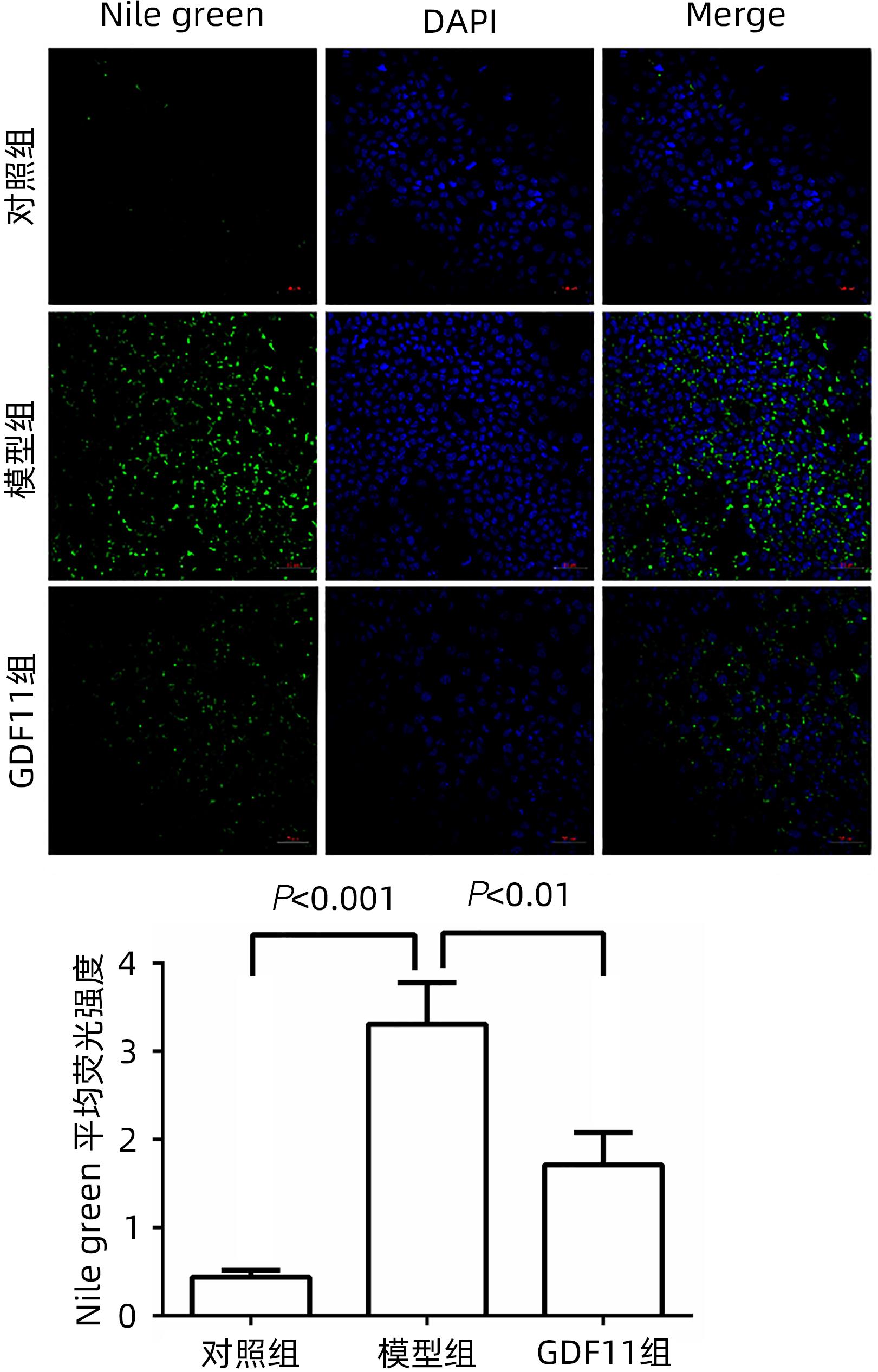
目的 探讨生长分化因子11(GDF11)对游离脂肪酸(FFA)诱导的肝细胞脂肪变性的影响,并探讨自噬在该过程中的作用。 方法 体外建立非酒精性脂肪性肝病(NAFLD)模型,加入重组GDF11,观察其对肝细胞脂肪变性及凋亡的影响;加入氯喹抑制自噬,探究GDF11对肝细胞自噬流的改变及其与脂肪变性的关系。AM-12细胞分为5组:对照组、对照+GDF11组、模型组、GDF11组和GDF11+氯喹组。对照组:DMEM完全培养基培养细胞;对照+GDF11组:在完全培养基的基础上加入GDF11(100 ng/mL);模型组:浓度为1 mmol/L的FFA(油酸∶棕榈酸=2∶1)诱导NAFLD模型;GDF11组:细胞中同时加入FFA和GDF11;GDF11+氯喹组:细胞中同时加入FFA、GDF11和浓度为20 μmol/L的氯喹(自噬抑制剂)。使用脂滴荧光染色和全自动生化分析仪观察肝细胞内脂质沉积情况,蛋白免疫印迹法及蛋白免疫荧光检测自噬相关蛋白(p62、LC3B)水平,观察细胞自噬水平;JC-1染色检测线粒体膜电位。多组间计量资料比较用单因素方差分析,进一步两两比较采用LSD-t检验。 结果 与模型组比较,GDF11组与对照组肝细胞内脂滴积累明显减少,脂滴体积明显减小(P值均<0.01)。模型组肝细胞内甘油三酯水平明显高于GDF11组和对照组(P值均<0.000 1)。模型组LC3B水平较对照组与GDF11组低(P值均<0.05),p62水平高(P值均<0.05);与GDF11组比较,GDF11+氯喹组细胞内甘油三酯含量升高(P<0.001);与模型组比较,GDF11组ROS含量明显减少(P<0.05)。FFA诱导过后,胞质中的绿色荧光明显升高,与对照组比较,模型组JC-1单体/多聚体升高(P<0.000 1),细胞线粒体膜电位下降;加入GDF11后,绿色荧光强度减弱,与模型组比较,GDF11组JC-1单体/多聚体下降(P<0.05),线粒体膜电位回升。 结论 GDF11可以改善FFA诱导的脂毒性肝损伤,并通过促进细胞自噬来改善NAFLD中肝细胞脂肪变性。 Abstract:Objective To investigate the effect of growth differentiation factor 11 (GDF11) on free fatty acid (FFA)-induced hepatocyte steatosis and the role of autophagy in this process. Methods A model of nonalcoholic fatty liver disease was established invitro, and recombinant GDF11 was added to observe its effect on hepatocyte steatosis and apoptosis. Chloroquine was added to inhibit autophagy, and the change in hepatocyte autophagy flow induced by GDF11 and its association with steatosis were explored. AM-12 cells were divided into control group, control+GDF11 group, model group, GDF11 group, and GDF11+chloroquine group. The cells in the control group were cultured with DMEM complete culture medium; the cells in the control+GDF11 group were cultured with GDF11 (100 ng/mL) added to the complete culture medium; the cells in the model group were treated with 1 mmol/L FFA (with an oleic acid/palmitic acid ratio of 2∶1) to induce hepatocyte steatosis; the cells in the GDF11 group were co-cultured with FFA and GDF11; the cells in the GDF11+chloroquine group were co-cultured with FFA, GDF11, and 20 μmol/L chloroquine (an autophagy inhibitor). Lipid droplet fluorescence staining and an automatic biochemical analyzer were used to observe lipid deposition in hepatocytes; Western blotting and protein immunofluorescence assay were used to measure the levels of autophagy-related proteins (p62 and LC3B) and the level of autophagy; JC-1 staining was used to measure mitochondrial membrane potential. A one-way analysis of variance was used for comparison of continuous data between multiple groups, and the least significant difference t-test was used for further comparison between two groups. Results Compared with the model group, the GDF11 group and the control group had significant reductions in the accumulation and volume of lipid droplets in hepatocytes (all P<0.05). The model group had a significantly higher level of triglyceride in hepatocytes than the GDF11 group and the control group (P<0.000 1). Compared with the control group, the model group had a significantly lower level of LC3B (P<0.05) and a significantly higher level of p62 (P<0.05); compared with the GDF11 group, the GDF11+chloroquine group had a significant increase in the level of triglyceride in hepatocytes (P<0.001); compared with the model group, the GDF11 group had a significant reduction in the content of ROS (P<0.05). After FFA induction, there was a significant increase in green fluorescence in cytoplasma, and compared with the control group, the model group had a significant increase in JC-1 monomers/aggregates (P<0.000 1) and a reduction in mitochondrial membrane potential; there was a reduction in the intensity of green fluorescence after the addition of GDF11, and compared with the model group, the GDF11 group had a significant reduction in JC-1 monomers/aggregates (P<0.05) and an increase in mitochondrial membrane potential. Conclusion GDF11 can alleviate FFA-induced lipotoxic liver injury and improve hepatocyte steatosis in nonalcoholic fatty liver disease by promoting autophagy. -
-
[1] POWELL EE, WONG VWS, RINELLA M. Non-alcoholic fatty liver disease[J]. Lancet, 2021, 397( 10290): 2212- 2224. DOI: 10.1016/S0140-6736(20)32511-3. [2] RUISSEN MM, MAK AL, BEUERS U, et al. Non-alcoholic fatty liver disease: A multidisciplinary approach towards a cardiometabolic liver disease[J]. Eur J Endocrinol, 2020, 183( 3): R57-R73. DOI: 10.1530/EJE-20-0065. [3] TANAKA N, KIMURA T, FUJIMORI N, et al. Current status, problems, and perspectives of non-alcoholic fatty liver disease research[J]. World J Gastroenterol, 2019, 25( 2): 163- 177. DOI: 10.3748/wjg.v25.i2.163. [4] LOOMBA R, FRIEDMAN SL, SHULMAN GI. Mechanisms and disease consequences of nonalcoholic fatty liver disease[J]. Cell, 2021, 184( 10): 2537- 2564. DOI: 10.1016/j.cell.2021.04.015. [5] GLUCHOWSKI NL, BECUWE M, WALTHER TC, et al. Lipid droplets and liver disease: From basic biology to clinical implications[J]. Nat Rev Gastroenterol Hepatol, 2017, 14( 6): 343- 355. DOI: 10.1038/nrgastro.2017.32. [6] KRAHMER N, FARESE RV Jr, WALTHER TC. Balancing the fat: Lipid droplets and human disease[J]. EMBO Mol Med, 2013, 5( 7): 973- 983. DOI: 10.1002/emmm.201100671. [7] MORTEZAEE K, KHANLARKHANI N. Melatonin application in targeting oxidative-induced liver injuries: A review[J]. J Cell Physiol, 2018, 233( 5): 4015- 4032. DOI: 10.1002/jcp.26209. [8] LINHART KB, GLASSEN K, PECCERELLA T, et al. The generation of carcinogenic etheno-DNA adducts in the liver of patients with nonalcoholic fatty liver disease[J]. Hepatobiliary Surg Nutr, 2015, 4( 2): 117- 123. DOI: 10.3978/j.issn.2304-3881.2015.01.14. [9] LIU Q, BENGMARK S, QU S. The role of hepatic fat accumulation in pathogenesis of non-alcoholic fatty liver disease(NAFLD)[J]. Lipds Heath Dis, 2010, 9( 1): 1- 9. DOI: 10.1186/1476-511X-9-42. [10] WOBSER H, DORN C, WEISS TS, et al. Lipid accumulation in hepatocytes induces fibrogenic activation of hepatic stellate cells[J]. Cell Res, 2009, 19( 8): 996- 1005. DOI: 10.1038/cr.2009.73. [11] BRAY F, FERLAY J, SOERJOMATARAM I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2018, 68( 6): 394- 424. DOI: 10.3322/caac.21492. [12] WALKER RG, POGGIOLI T, KATSIMPARDI L, et al. Biochemistry and biology of GDF11 and myostatin: Similarities, differences, and questions for future investigation[J]. Circ Res, 2016, 118( 7): 1125- 1141. DOI: 10.1161/CIRCRESAHA.116.308391. [13] LU BX, ZHONG JN, PAN JF, et al. Gdf11 gene transfer prevents high fat diet-induced obesity and improves metabolic homeostasis in obese and STZ-induced diabetic mice[J]. J Transl Med, 2019, 17( 1): 422. DOI: 10.1186/s12967-019-02166-1. [14] FROHLICH J, KOVACOVICOVA K, MAZZA T, et al. GDF11 induces mild hepatic fibrosis independent of metabolic health[J]. Aging, 2020, 12( 20): 20024- 20046. DOI: 10.18632/aging.104182. [15] ZHOU J, PANG J, TRIPATHI M, et al. Spermidine-mediated hypusination of translation factor EIF5A improves mitochondrial fatty acid oxidation and prevents non-alcoholic steatohepatitis progression[J]. Nat Commun, 2022, 13( 1): 5202. DOI: 10.1038/s41467-022-32788-x. [16] GADIPARTHI C, SPATZ M, GREENBERG S, et al. NAFLD epidemiology, emerging pharmacotherapy, liver transplantation implications and the trends in the United States[J]. J Clin Transl Hepatol, 2020, 8( 2): 215- 221. DOI: 10.14218/JCTH.2020.00014. [17] BYRNES K, BLESSINGER S, BAILEY NT, et al. Therapeutic regulation of autophagy in hepatic metabolism[J]. Acta Pharm Sin B, 2022, 12( 1): 33- 49. DOI: 10.1016/j.apsb.2021.07.021. [18] ZHONG CC, ZHAO T, HOGSTRAND C, et al. Copper(Cu) induced changes of lipid metabolism through oxidative stress-mediated autophagy and Nrf2/PPARγ pathways[J]. J Nutr Biochem, 2022, 100: 108883. DOI: 10.1016/j.jnutbio.2021.108883. [19] SCORLETTI E, CARR RM. A new perspective on NAFLD: Focusing on lipid droplets[J]. J Hepatol, 2022, 76( 4): 934- 945. DOI: 10.1016/j.jhep.2021.11.009. [20] LIN CW, ZHANG H, LI M, et al. Pharmacological promotion of autophagy alleviates steatosis and injury in alcoholic and non-alcoholic fatty liver conditions in mice[J]. J Hepatol, 2013, 58( 5): 993- 999. DOI: 10.1016/j.jhep.2013.01.011. [21] DAI Z, SONG GQ, BALAKRISHNAN A, et al. Growth differentiation factor 11 attenuates liver fibrosis via expansion of liver progenitor cells[J]. Gut, 2020, 69( 6): 1104- 1115. DOI: 10.1136/gutjnl-2019-318812. [22] JIAO L, SHAO YC, YU Q, et al. GDF11 replenishment protects against hypoxia-mediated apoptosis in cardiomyocytes by regulating autophagy[J]. Eur J Pharmacol, 2020, 885: 173495. DOI: 10.1016/j.ejphar.2020.173495. -


 PDF下载 ( 1551 KB)
PDF下载 ( 1551 KB)

 下载:
下载: