| [1] |
CHEN W, ZHENG R, BAADE PD, et al. Cancer statistics in China, 2015[J]. CA Cancer J Clin, 2016, 66(2): 115-132. DOI: 10.3322/caac.21338. |
| [2] |
General Office of National Health Commission. Standard for diagnosis and treatment of pancreatic cancer (2022 edition)[J]. J Clin Hepatol, 2022, 38(5): 1006-1015. DOI: 10.3969/j.issn.1001-5256.2022.05.007. |
| [3] |
ZHU H, WEI M, XU J, et al. PARP inhibitors in pancreatic cancer: molecular mechanisms and clinical applications[J]. Mol Cancer, 2020, 19(1): 49. DOI: 10.1186/s12943-020-01167-9. |
| [4] |
HAN A, XU R, LIU Y, et al. HSDL2 acts as a promoter in pancreatic cancer by regulating cell proliferation and lipid metabolism[J]. Onco Targets Ther, 2021, 14: 435-444. DOI: 10.2147/OTT.S287722. |
| [5] |
NEOPTOLEMOS JP, KLEEFF J, MICHL P, et al. Therapeutic developments in pancreatic cancer: current and future perspectives[J]. Nat Rev Gastroenterol Hepatol, 2018, 15(6): 333-348. DOI: 10.1038/s41575-018-0005-x. |
| [6] |
HAO TY, HE WQ. Advances in metabolic engineering of macrolide antibiotics[J]. Chin J Biotech, 2021, 37(5): 1737-1747. DOI: 10.13345/j.cjb.200686. |
| [7] |
JIN Y, ZUO HX, LI MY, et al. Anti-tumor effects of carrimycin and monomeric isovalerylspiramycin I on hepatocellular carcinoma in vitro and in vivo[J]. Front Pharmacol, 2021, 12: 774231. DOI: 10.3389/fphar.2021.774231. |
| [8] |
LIANG SY, ZHAO TC, ZHOU ZH, et al. Anti-tumor effect of carrimycin on oral squamous cell carcinoma cells in vitro and in vivo[J]. Transl Oncol, 2021, 14(6): 101074. DOI: 10.1016/j.tranon.2021.101074. |
| [9] |
|
| [10] |
EDELMAN MJ, SHVARTSBEYN M. Epothilones in development for non—small-cell lung cancer: novel anti-tubulin agents with the potential to overcome taxane resistance[J]. Clin Lung Cancer, 2012, 13(3): 171-180. DOI: 10.1016/j.cllc.2011.02.005. |
| [11] |
GIANNAKAKOU P, GUSSIO R, NOGALES E, et al. A common pharmacophore for epothilone and taxanes: molecular basis for drug resistance conferred by tubulin mutations in human cancer cells[J]. Proc Natl Acad Sci U S A, 2000, 97(6): 2904-2909. DOI: 10.1073/pnas.040546297. |
| [12] |
LI YL, SUN J, HU X, et al. Epothilone B induces apoptosis and enhances apoptotic effects of ABT-737 on human cancer cells via PI3K/AKT/mTOR pathway[J]. J Cancer Res Clin Oncol, 2016, 142(11): 2281-2289. DOI: 10.1007/s00432-016-2236-y. |
| [13] |
CUI J, ZHOU J, HE W, et al. Targeting selenoprotein H in the nucleolus suppresses tumors and metastases by Isovalerylspiramycin I[J]. J Exp Clin Cancer Res, 2022, 41(1): 126. DOI: 10.1186/s13046-022-02350-0. |
| [14] |
LIU Y, QUAN Y. Relationship between inflammatory microenvironment and epithelial-mesenchymal transition and its significance in pancreatic carcinoma[J]. Chin J Clin Res, 2018, 31(12): 1619-1623. DOI: 10.13429/j.cnki.cjcr.2018.12.006. |
| [15] |
BAKIR B, CHIARELLA AM, PITARRESI JR, et al. EMT, MET, plasticity, and tumor metastasis[J]. Trends Cell Biol, 2020, 30(10): 764-776. DOI: 10.1016/j.tcb.2020.07.003. |
| [16] |
YEUNG KT, YANG J. Epithelial-mesenchymal transition in tumor metastasis[J]. Mol Oncol, 2017, 11(1): 28-39. DOI: 10.1002/1878-0261.12017. |
| [17] |
KONG HR, DAI SJ, CHEN H, et al. α-solanine inhibits epithelial-mesenchymal transition in trichostatin A-resistant human pancreatic cancer cells through the WNT signaling pathway[J]. J Hepatopancreatobiliary Surg, 2020, 32(2): 89-96. DOI: 10.11952/j.issn.1007-1954.2020.02.006. |
| [18] |
LIU JB, QI M, LI QQ, et al. The mechanism of cordycepin in inhibition of pancreatic cancer stem cells proliferation and metastasis[J]. Acta Pharm Sin, 2017, 52(9): 1404-1409. DOI: 10.16438/j.0513-4870.2017-0353. |


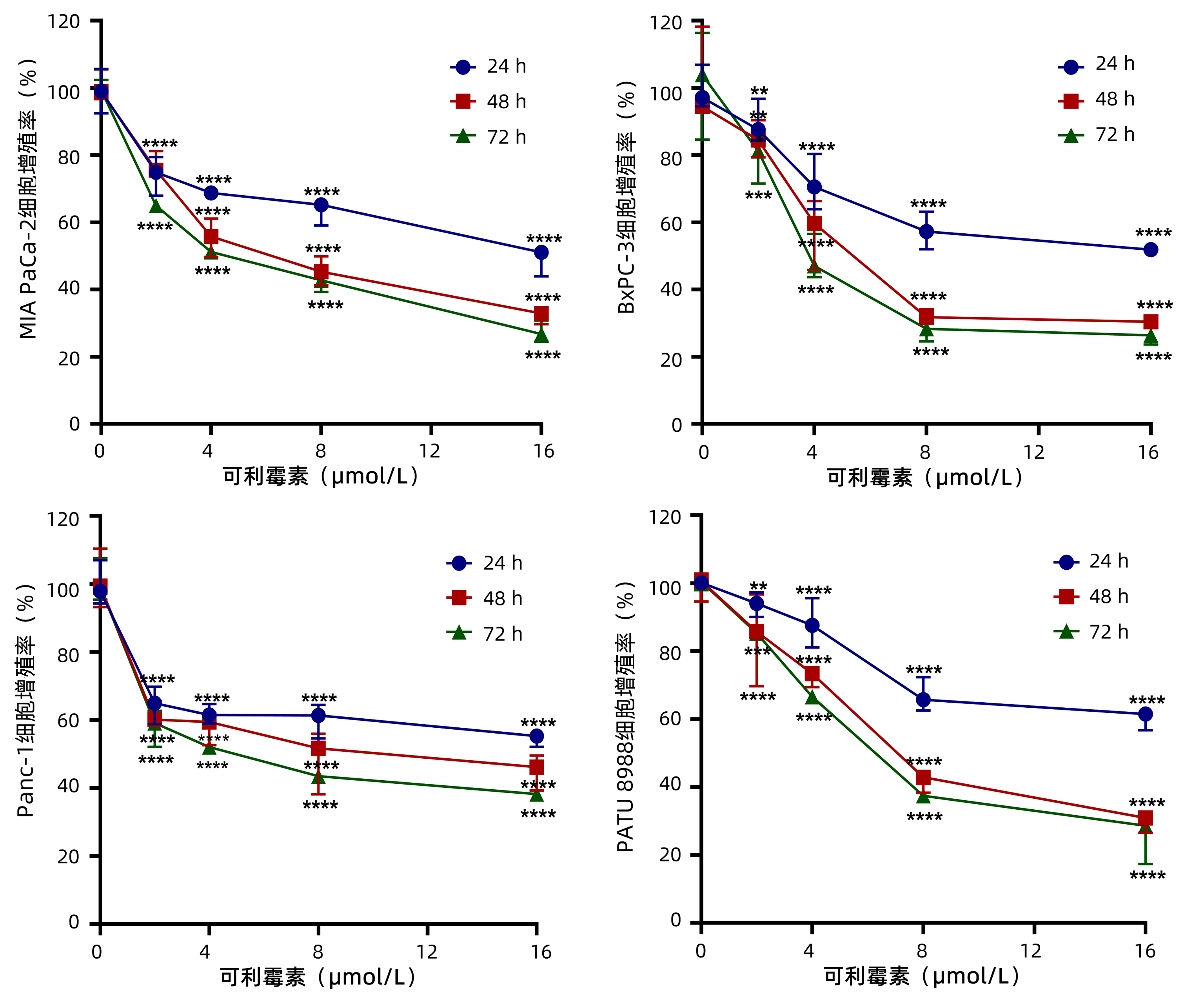




 DownLoad:
DownLoad:


