代谢相关脂肪性肝病患者肝纤维化标志物、炎症标志物与胆结石患病风险的关联性分析
DOI: 10.12449/JCH260312
Association of liver fibrosis markers and inflammation markers with the risk of gallstones in patients with metabolic dysfunction-associated fatty liver disease
-
摘要:
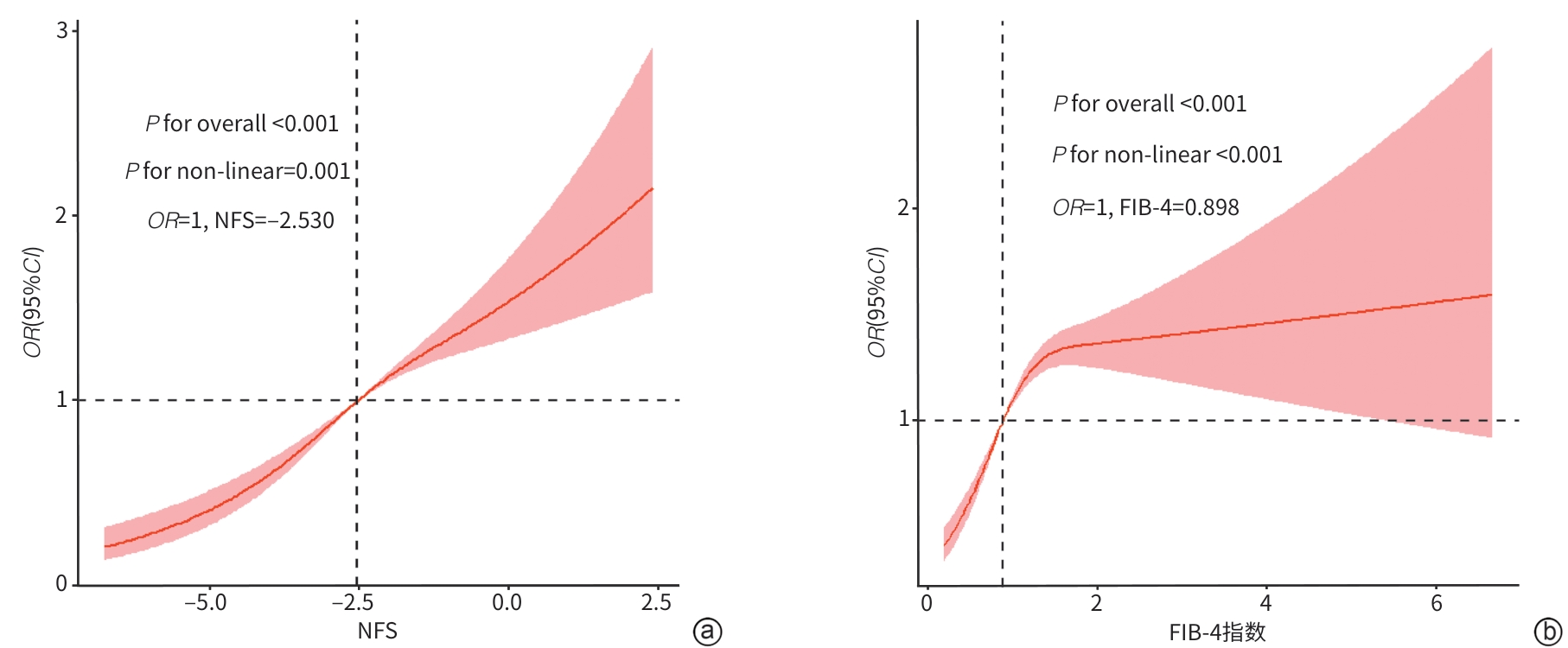
目的 阐明代谢相关脂肪性肝病(MAFLD)患者肝纤维化评分、炎症标志物与胆结石之间的关联,探索肝纤维化评分在炎症标志物与胆结石关系中的中介作用。 方法 回顾性纳入2014年1月—2023年6月在苏北人民医院接受体检并确诊MAFLD的患者,根据腹部彩超检查结果分为胆结石组(n=1 724)和非胆结石组(n=12 843)。收集患者临床数据,包括人口统计学信息、相关病史和家族史,以及体格检查、彩色多普勒超声检查和实验室生化指标。与代谢紊乱和胰岛素抵抗相关的生物标志物包括甘油三酯-葡萄糖指数(TyG)、TyG-体重指数(TyG-BMI)、血浆致动脉粥样硬化指数(AIP)及非高密度脂蛋白胆固醇与高密度脂蛋白胆固醇比值(NHHR);与炎症和营养状况相关的生物标志物包括中性粒细胞与淋巴细胞比值(NLR)、中性粒细胞百分比与白蛋白比值(NPAR)及单核细胞与淋巴细胞比值(MLR);评估肝纤维化程度和肝功能的生物标志物包括白蛋白-胆红素(ALBI)评分、非酒精性脂肪性肝病纤维化评分(NFS)、纤维化-4(FIB-4)指数和天冬氨酸氨基转移酶与血小板比值指数(APRI)。符合正态分布的计量资料2组间比较采用成组t检验;非正态分布的计量资料2组间比较采用Mann-Whitney U检验;计数资料2组间比较采用χ2检验。采用多因素Logistic回归分析、限制性立方样条分析和中介效应分析研究肝纤维化标志物、炎症标志物与胆结石患病风险之间的关联。 结果 MAFLD患者的胆结石患病率为11.8%。胆结石组与非胆结石组在性别、年龄、吸烟史、糖尿病、高血压、淋巴细胞、血小板、葡萄糖、白蛋白、血清尿酸、丙氨酸氨基转移酶、天冬氨酸氨基转移酶、红细胞、NLR、NPAR、MLR、NFS、FIB-4指数和ALBI评分方面均存在显著差异(P值均<0.05)。多因素Logistic回归分析结果显示,在完全调整后的模型中,NLR[比值比(odds,ratio,OR)=1.091,95%置信区间(95% confidence interval, 95%CI):1.028~1.160]、NPAR(OR=1.073,95%CI:1.042~1.105)、MLR(OR=1.142,95%CI:1.057~1.232)、NFS(OR=1.239,95%CI:1.190~1.291)和FIB-4指数(OR=1.326,95%CI:1.241~1.417)是胆结石患病风险的独立影响因素(P值均<0.05)。限制性立方样条分析显示,NFS和FIB-4指数与胆结石发生风险之间存在显著的非线性关联(非线性P值均<0.05)。中介分析进一步表明,NLR、MLR和NPAR与胆结石的关联部分可通过NFS或FIB-4指数介导,NFS和FIB-4指数的中介占比分别为36.79%、28.09%、29.67%和18.31%、17.70%、11.57%。 结论 MAFLD患者中,NFS和FIB-4指数与胆结石的患病率呈非线性关联,且介导炎症标志物NLR、NPAR和MLR与胆结石风险之间的关联。 Abstract:Objective To investigate the association of liver fibrosis scores and inflammation markers with gallstones in patients with metabolic dysfunction-associated fatty liver disease (MAFLD), as well as the mediating role of liver fibrosis scores in the relationship between inflammation markers and gallstones. Methods A total of 14 567 patients who received physical examination and were diagnosed with MAFLD in Subei People’s Hospital from January 2014 to June 2023 were enrolled in this study, and according to the results of abdominal color Doppler ultrasound, they were divided into gallstone group with 1 724 patients and non-gallstone group with 12 843 patients. Related clinical data were collected from all patients, including demographic data, medical history, family history, physical examination, Color Doppler ultrasound, and biochemical parameters. The biomarkers associated with metabolic disorders and insulin resistance included triglyceride-glucose index (TyG), TyG-body mass index (BMI) index, atherogenic index of plasma (AIP), and non-high-density lipoprotein cholesterol-to-high-density lipoprotein cholesterol ratio (NHHR); the biomarkers associated with inflammation and nutritional status included neutrophil-to-lymphocyte ratio (NLR), neutrophil percentage-to-albumin ratio (NPAR), and monocyte-to-lymphocyte ratio (MLR); the biomarkers for assessing liver fibrosis degree and liver function included albumin-bilirubin (ALBI) score, NAFLD fibrosis score (NFS), fibrosis-4 (FIB-4) index, and aspartate aminotransferase-to-platelet ratio index (APRI). The independent-samples t test was used for comparison of normally distributed continuous data between two groups, while the Mann-Whitney U test was used for comparison of non-normally distributed continuous data between two groups; the chi-square test was used for comparison of categorical data between two groups. Multivariate Logistic regression analysis, restricted cubic spline analysis, and mediating effect analysis were used to assess the association of liver fibrosis markers and inflammation markers with the risk of gallstones. Results The prevalence rate of gallstones was 11.8% among the MAFLD patients. There were significant differences between the gallstone group and the non-gallstone group in sex, age, smoking history, diabetes, hypertension, lymphocytes, platelets, glucose, albumin, serum uric acid, alanine aminotransferase, aspartate aminotransferase, red blood cell, NLR, NPAR, MLR, NFS, FIB-4 index, and ALBI score (all P<0.05). The multivariate Logistic regression analysis showed that NLR (odds ratio [OR]=1.091, 95% confidence interval [CI]: 1.028 — 1.160, P<0.05), NPAR (OR=1.073, 95%CI: 1.042 — 1.105, P<0.05), MLR (OR=1.142, 95%CI: 1.057 — 1.232, P<0.05), NFS (OR=1.239, 95%CI: 1.190 — 1.291, P<0.05), and FIB-4 index (OR=1.326, 95%CI: 1.241 — 1.417, P<0.05) were influencing factors for the prevalence rate of gallstones. The restricted cubic spline analysis showed a significant non-linear association between NFS/FIB-4 index and the risk of gallstone (non-linear P<0.05). The mediating effect analysis further showed that the association of NLR, MLR, and NPAR with gallstones was partially mediated by NFS or FIB-4 index, with a mediating effect accounting for 36.79%、28.09%、29.67% and 18.31%、17.70、11.57%, respectively. Conclusion NFS and FIB-4 index have a non-linear association with the prevalence rate of gallstones in MAFLD patients, and they also mediate the association of NLR, NPAR, and MLR with the risk of gallstone. -
表 1 MAFLD患者胆结石组与非胆结石组基线资料
Table 1. Comparison of baseline characteristics between gallstone group and non-gallstone group in MAFLD patients
项目 胆结石组(n=1 724) 非胆结石组(n=12 843) 统计值 P值 性别[例(%)] χ2=27.371 <0.001 男 1 213(70.4) 9 778(76.1) 女 511(29.6) 3 065(23.9) 年龄(岁) 53.48±12.88 47.55±13.14 t=17.877 <0.001 BMI(kg/m2) 26.62±2.91 26.61±3.04 t=0.159 0.872 吸烟史[例(%)] 381(22.1) 2 238(17.4) χ2=22.517 <0.001 糖尿病[例(%)] 518(30.0) 2 926(22.8) χ2=44.421 <0.001 高血压[例(%)] 474(27.5) 3 010(23.4) χ2=13.751 <0.001 WBC(×109/L) 6.61±1.58 6.65±1.61 t=-0.873 0.382 NE(×109/L) 3.97±1.17 3.91±1.20 t=1.955 0.051 L(×109/L) 2.06±0.61 2.15±0.62 t=-5.758 <0.001 M(×109/L) 0.39±0.12 0.39±0.11 t=0.816 0.414 PLT(×109/L) 225.30±61.09 232.46±59.80 t=-4.581 <0.001 TC(mmol/L) 4.84±0.99 4.84±0.95 t=0.179 0.858 LDL-C(mmol/L) 3.03±0.82 3.02±0.81 t=0.435 0.663 HDL-C(mmol/L) 1.12±0.25 1.11±0.26 t=1.488 0.137 TG(mmol/L) 1.86(1.39~2.63) 1.90(1.38~2.78) Z=-1.574 0.116 Glu(mmol/L) 6.01±1.83 5.73±1.65 t=5.981 <0.001 Alb(g/L) 46.54±2.36 46.95±2.39 t=-6.816 <0.001 TP(g/L) 73.89±3.88 73.87±3.81 t=0.166 0.867 SUA(μmol/L) 372.28±84.43 365.69±85.14 t=3.039 0.002 Scr(μmol/L) 83.15±17.13 82.77±14.61 t=0.891 0.372 ALT(U/L) 24.00(17.00~35.00) 26.00(18.00~40.00) Z=-6.354 <0.001 AST(U/L) 21.00(17.00~26.00) 22.00(18.00~28.00) Z=-5.705 <0.001 TBil(μmol/L) 11.10(8.60~14.40) 11.00(8.40~14.40) Z=1.912 0.056 DBil(μmol/L) 4.00(3.20~5.00) 3.90(3.10~5.00) Z=0.458 0.647 RBC(×1012/L) 4.99±0.48 5.06±0.47 t=-5.396 <0.001 TyG 9.82±0.63 9.79±0.64 t=1.301 0.193 TyG-BMI 261.48±34.19 260.83±35.54 t=0.736 0.462 AIP 0.52(0.14~0.97) 0.56(0.14~1.04) Z=1.919 0.055 NHHR 3.38(2.70~4.16) 3.35(2.70~4.05) Z=1.275 0.202 NLR 2.05±0.78 1.93±0.81 t=5.994 <0.001 NPAR 12.85±1.73 12.46±1.74 t=8.644 <0.001 MLR 0.198±0.065 0.188±0.063 t=5.936 <0.001 NFS -2.15(-2.98~-1.21) -2.58(-3.41~-1.67) Z=12.939 <0.001 FIB-4指数 1.03(0.74~1.47) 0.88(0.61~1.27) Z=11.681 <0.001 APRI 0.24(0.18~0.32) 0.24(0.19~0.33) Z=1.597 0.124 ALBI评分 -3.26±0.21 -3.30±0.22 t=6.893 <0.001 注:MAFLD,代谢相关脂肪性肝病;BMI,体重指数;SUA,血清尿酸;L,淋巴细胞;M,单核细胞;NE,中性粒细胞;PLT,血小板;RBC,红细胞;Alb,白蛋白;Scr,血清肌酐;TG,甘油三酯;Glu,葡萄糖;LDL-C,低密度脂蛋白胆固醇;HDL-C,高密度脂蛋白胆固醇;TBil,总胆红素;AST,天冬氨酸氨基转移酶;ALT,丙氨酸氨基转移酶;ALBI,白蛋白-胆红素;FIB-4,纤维化-4;NFS,非酒精性脂肪性肝病纤维化评分;APRI,天冬氨酸氨基转移酶与血小板比值指数;NLR,中性粒细胞与淋巴细胞比值;NPAR,中性粒细胞百分比与白蛋白比值;AIP,血浆致动脉粥样硬化指数;TyG,甘油三酯-葡萄糖指数;NHHR,非高密度脂蛋白胆固醇与高密度脂蛋白胆固醇比值;MLR,单核细胞与淋巴细胞比值;TP,总蛋白;WBC,白细胞;TC,总胆固醇;DBil,直接胆红素。
表 2 多因素Logistic回归分析结果
Table 2. Multivariate Logistic regression analysis results
变量 模型1 模型2 模型3 OR(95%CI) P值 OR(95%CI) P值 OR(95%CI) P值 NLR 1.168(1.104~1.237) <0.001 1.097(1.037~1.161) 0.001 1.091(1.028~1.160) 0.002 NPAR 1.134(1.102~1.167) <0.001 1.079(1.046~1.114) <0.001 1.073(1.042~1.105) <0.001 MLR 1.251(1.162~1.345) <0.001 1.152(1.065~1.244) <0.001 1.142(1.057~1.232) <0.001 ALBI评分 1.084(1.059~1.110) <0.001 1.022(0.992~1.053) 0.157 1.011(0.986~1.037) 0.375 NFS 1.279(1.227~1.334) <0.001 1.260(1.213~1.308) <0.001 1.239(1.190~1.291) <0.001 FIB-4指数 1.367(1.281~1.459) <0.001 1.346(1.252~1.447) <0.001 1.326(1.241~1.417) <0.001 注:FIB-4,纤维化-4;NFS,非酒精性脂肪性肝病纤维化评分;ALBI,白蛋白-胆红素;NLR,中性粒细胞与淋巴细胞比值;NPAR,中性粒细胞百分比与白蛋白比值;MLR,单核细胞与淋巴细胞比值;OR,比值比;95%CI,95%置信区间。
表 3 NFS与FIB-4指数在炎症指标与胆结石关联中的中介效应
Table 3. Mediating effects of NFS and FIB-4 indices on the association between inflammatory markers and gallstones
中介变量 平均因果中介效应量(95%CI) 平均直接效应量(95%CI) 总效应量(95%CI) 中介占比1)(95%CI) P值 NPAR NFS 0.001(0.001~0.002) 0.002(0.002~0.003) 0.003(0.003~0.005) 29.67(21.42~41.06) <0.001 FIB-4指数 0.000(0.000~0.001) 0.003(0.003~0.004) 0.003(0.003~0.005) 11.57(7.76~17.23) <0.001 NLR NFS 0.005(0.004~0.007) 0.008(0.003~0.014) 0.013(0.009~0.021) 36.79(24.91~66.22) <0.001 FIB-4指数 0.003(0.002~0.004) 0.011(0.007~0.020) 0.013(0.009~0.021) 18.31(11.92~30.03) <0.001 MLR NFS 0.110(0.087~0.141) 0.285(0.139~0.442) 0.395(0.237~0.556) 28.09(18.76~43.42) <0.001 FIB-4指数 0.069(0.050~0.091) 0.322(0.173~0.482) 0.391(0.240~0.551) 17.70(10.44~29.12) <0.001 注:NPAR,中性粒细胞百分比与白蛋白比值;NFS,非酒精性脂肪性肝病纤维化评分;FIB-4,纤维化-4;NLR,中性粒细胞与淋巴细胞比值;MLR,单核细胞与淋巴细胞比值;95%CI,95%置信区间。1)中介占比=平均因果中介效应/总效应×100%。
-
[1] RINELLA ME, LAZARUS JV, RATZIU V, et al. A multisociety Delphi consensus statement on new fatty liver disease nomenclature[J]. J Hepatol, 2023, 79( 6): 1542- 1556. DOI: 10.1016/j.jhep.2023.06.003. [2] KOYAMA Y, BRENNER DA. Liver inflammation and fibrosis[J]. J Clin Invest, 2017, 127( 1): 55- 64. DOI: 10.1172/JCI88881. [3] LU YN, XU XL, WU JL, et al. Association between neutrophil-to-high-density lipoprotein cholesterol ratio and metabolic dysfunction-associated steatotic liver disease and liver fibrosis in the US population: A nationally representative cross-sectional study using NHANES data from 2017 to 2020[J]. BMC Gastroenterol, 2024, 24( 1): 300. DOI: 10.1186/s12876-024-03394-6. [4] LIU ZY, ZHUANG YJ, DONG X, et al. Construction of a non-invasive diagnostic model for liver fibrosis in non-alcoholic fatty liver disease[J]. Chin J Med Offic, 2024, 52( 7): 688- 691. DOI: 10.16680/j.1671-3826.2024.07.07.刘正一, 庄颖洁, 董旭, 等. 非酒精性脂肪性肝病肝纤维化无创诊断模型构建[J]. 临床军医杂志, 2024, 52( 7): 688- 691. DOI: 10.16680/j.1671-3826.2024.07.07. [5] PAN L, WANG LX, MA HJ, et al. Relevance of combined influence of nutritional and inflammatory status on non-alcoholic fatty liver disease and advanced fibrosis: A mediation analysis of lipid biomarkers[J]. J Gastroenterol Hepatol, 2024, 39( 12): 2853- 2862. DOI: 10.1111/jgh.16760. [6] ZHAO XY, WANG N, SUN YY, et al. Screen-detected gallstone disease and risk of liver and pancreatic cancer: The Kailuan Cohort Study[J]. Liver Int, 2020, 40( 7): 1744- 1755. DOI: 10.1111/liv.14456. [7] WANG YK, SHANG XC, ZHANG YC, et al. The association between neutrophil to high-density lipoprotein cholesterol ratio and gallstones: A cross-sectional study[J]. BMC Public Health, 2025, 25( 1): 157. DOI: 10.1186/s12889-025-21392-5. [8] GONG L, FAN SJ, PENG ZF, et al. The association between triglyceride-glucose index and gallstones: NHANES 2017-2020[J]. Front Endocrinol, 2024, 15: 1437711. DOI: 10.3389/fendo.2024.1437711. [9] ZHANG YY, ZHANG JJ, YU D, et al. Association between metabolic dysfunction associated steatotic liver disease and gallstones in the US population using propensity score matching[J]. Sci Rep, 2025, 15( 1): 910. DOI: 10.1038/s41598-025-85218-5. [10] DAWOD S, BROWN K. Non-invasive testing in metabolic dysfunction-associated steatotic liver disease[J]. Front Med, 2024, 11: 1499013. DOI: 10.3389/fmed.2024.1499013. [11] CHAN WK, CHUAH KH, RAJARAM RB, et al. Metabolic dysfunction-associated steatotic liver disease(MASLD): A state-of-the-art review[J]. J Obes Metab Syndr, 2023, 32( 3): 197- 213. DOI: 10.7570/jomes23052. [12] ZENG YL, LIN L, CHEN JL, et al. Prognostic value of glycolipid metabolism index on complications and mechanical ventilation in intensive care unit patients with intracerebral hemorrhage: A retrospective cohort study using the MIMIC-IV database[J]. Front Neurol, 2025, 16: 1516627. DOI: 10.3389/fneur.2025.1516627. [13] ZAWIAH M, KHAN AH, FARHA R ABU, et al. Predictors of stroke-associated pneumonia and the predictive value of neutrophil percentage-to-albumin ratio[J]. Postgrad Med, 2023, 135( 7): 681- 689. DOI: 10.1080/00325481.2023.2261354. [14] HYDES TJ, KENNEDY OJ, GLYN-OWEN K, et al. Liver fibrosis assessed via noninvasive tests is associated with incident heart failure in a general population cohort[J]. Clin Gastroenterol Hepatol, 2024, 22( 8): 1657- 1667. DOI: 10.1016/j.cgh.2024.03.045. [15] TOYODA H, JOHNSON PJ. The ALBI score: From liver function in patients with HCC to a general measure of liver function[J]. JHEP Rep, 2022, 4( 10): 100557. DOI: 10.1016/j.jhepr.2022.100557. [16] LUO S, WENG XL, XU J, et al. Correlation between ZJU index and hepatic steatosis and liver fibrosis in American adults with NAFLD[J]. Front Med, 2024, 11: 1443811. DOI: 10.3389/fmed.2024.1443811. [17] ACALOVSCHI M. Gallstones in patients with liver cirrhosis: Incidence, etiology, clinical and therapeutical aspects[J]. World J Gastroenterol, 2014, 20( 23): 7277- 7285. DOI: 10.3748/wjg.v20.i23.7277. [18] MALLICK B, ANAND AC. Gallstone disease in cirrhosis: Pathogenesis and management[J]. J Clin Exp Hepatol, 2022, 12( 2): 551- 559. DOI: 10.1016/j.jceh.2021.09.011. [19] KUL K, SERIN E, YAKAR T, et al. Autonomic neuropathy and gallbladder motility in patients with liver cirrhosis[J]. Turk J Gastroenterol, 2015, 26( 3): 254- 258. DOI: 10.5152/tjg.2015.4469. [20] KANWAL F, KRAMER JR, LI L, et al. GLP-1 receptor agonists and risk for cirrhosis and related complications in patients with metabolic dysfunction-associated steatotic liver disease[J]. JAMA Intern Med, 2024, 184( 11): 1314- 1323. DOI: 10.1001/jamainternmed.2024.4661. [21] JIANG TT, DENG W, WANG SY, et al. Diagnosis and evaluation of fatty liver disease[J/OL]. Chin J Liver Dis(Electronic Version), 2024, 16( 2): 22- 28. DOI: 10.3969/j.issn.1674-7380.2024.02.005.蒋婷婷, 邓雯, 王诗雨, 等. 脂肪性肝病的诊断和评估[J/OL]. 中国肝脏病杂志(电子版), 2024, 16( 2): 22- 28. DOI: 10.3969/j.issn.1674-7380.2024.02.005. [22] FU C, CHEN JH, WANG YX, et al. Association between complete blood cell count-derived inflammatory biomarkers and gallstones prevalence in American adults under 60 years of age[J]. Front Immunol, 2024, 15: 1497068. DOI: 10.3389/fimmu.2024.1497068. [23] WANG JJ, SHEN ZW, LIANG YW, et al. Association of neutrophil percentage to albumin ratio with gallstones: A cross-sectional study from the United States NHANES[J]. BMC Public Health, 2024, 24( 1): 3503. DOI: 10.1186/s12889-024-21071-x. [24] WANG HH, PORTINCASA P, MENDEZ-SANCHEZ N, et al. Effect of ezetimibe on the prevention and dissolution of cholesterol gallstones[J]. Gastroenterology, 2008, 134( 7): 2101- 2110. DOI: 10.1053/j.gastro.2008.03.011. [25] MAURER KJ, CAREY MC, FOX JG. Roles of infection, inflammation, and the immune system in cholesterol gallstone formation[J]. Gastroenterology, 2009, 136( 2): 425- 440. DOI: 10.1053/j.gastro.2008.12.031. [26] KÄRKKÄINEN J, ASPINEN S, HARJU J, et al. Plasma glutathione peroxidase(GPX1) levels and oxidative stress in gallstone patients operated with two different cholecystectomy techniques: A randomized study with special reference to cancer patients[J]. Anticancer Res, 2017, 37( 12): 6921- 6927. DOI: 10.21873/anticanres.12156. [27] JIANG GH, LI S, LI HY, et al. Bidirectional associations among gallstone disease, non-alcoholic fatty liver disease, kidney stone disease[J]. World J Gastroenterol, 2024, 30( 46): 4914- 4928. DOI: 10.3748/wjg.v30.i46.4914. [28] ZHANG S, LIU J, SHI XJ, et al. Association between triglyceride-glucose index and gallstones in women: A cross-sectional study[J]. J Clin Hepatol, 2025, 41( 7): 1407- 1413. DOI: 10.12449/JCH250727.章帅, 刘军, 史茜菁, 等. 甘油三酯-葡萄糖(TyG)指数与女性胆囊结石的相关性分析[J]. 临床肝胆病杂志, 2025, 41( 7): 1407- 1413. DOI: 10.12449/JCH250727. [29] WANG JJ, LI H, HU JC, et al. Relationship of triglyceride-glucose index to gallstone prevalence and age at first gallstone surgery in American adults[J]. Sci Rep, 2024, 14( 1): 16749. DOI: 10.1038/s41598-024-67883-0. [30] LAM SM, WANG ZH, SONG JW, et al. Non-invasive lipid panel of MASLD fibrosis transition underscores the role of lipoprotein sulfatides in hepatic immunomodulation[J]. Cell Metab, 2025, 37( 1): 69- 86.e7. DOI: 10.1016/j.cmet.2024.09.009. [31] JUAN CA, PÉREZ DE LA LASTRA JM, PLOU FJ, et al. The chemistry of reactive oxygen species(ROS) revisited: Outlining their role in biological macromolecules(DNA, lipids and proteins) and induced pathologies[J]. Int J Mol Sci, 2021, 22( 9): 4642. DOI: 10.3390/ijms22094642. [32] MA M, JIANG W, ZHOU RB. DAMPs and DAMP-sensing receptors in inflammation and diseases[J]. Immunity, 2024, 57( 4): 752- 771. DOI: 10.1016/j.immuni.2024.03.002. -



 PDF下载 ( 1257 KB)
PDF下载 ( 1257 KB)

 下载:
下载:


