司曲替尼对四氯化碳诱导的肝纤维化小鼠模型的影响及其作用机制
DOI: 10.12449/JCH260315
Effect of sitravatinib on a mouse model of carbon tetrachloride-induced liver fibrosis and its mechanism
-
摘要:
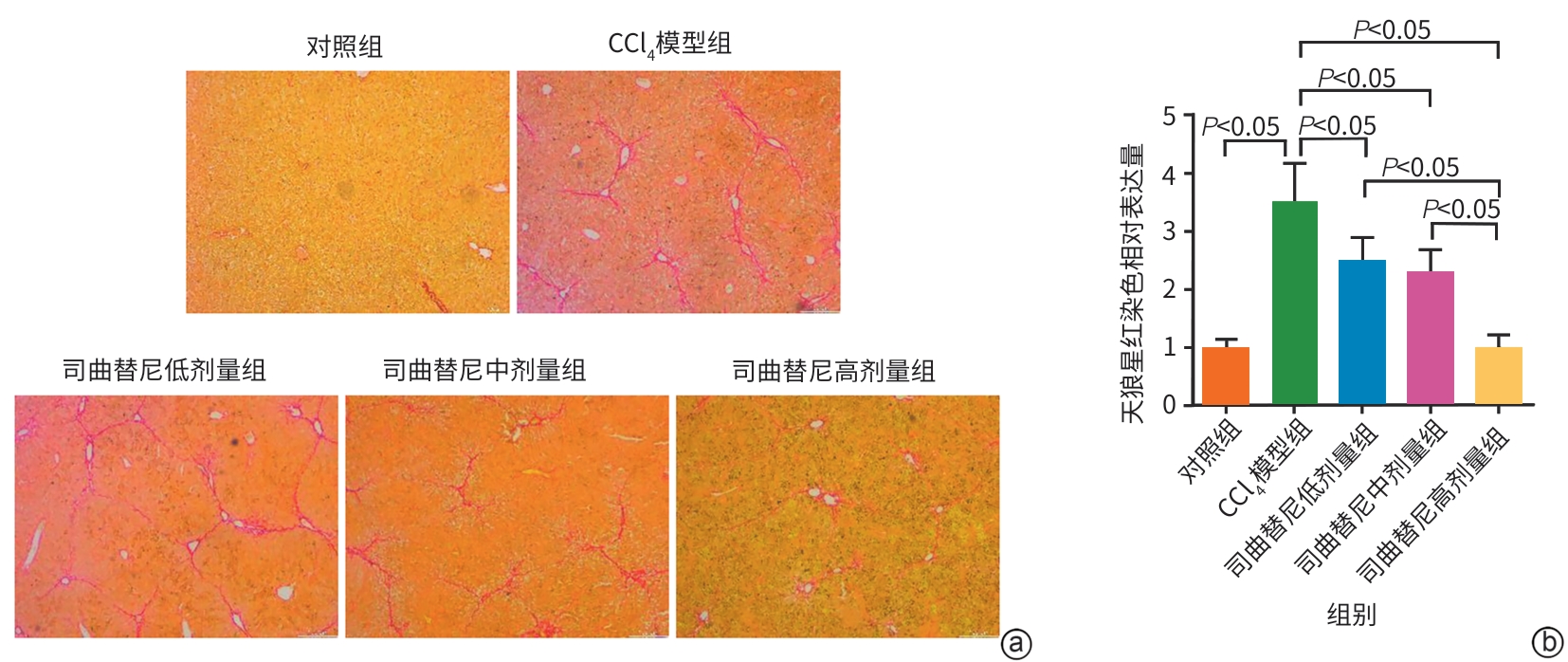
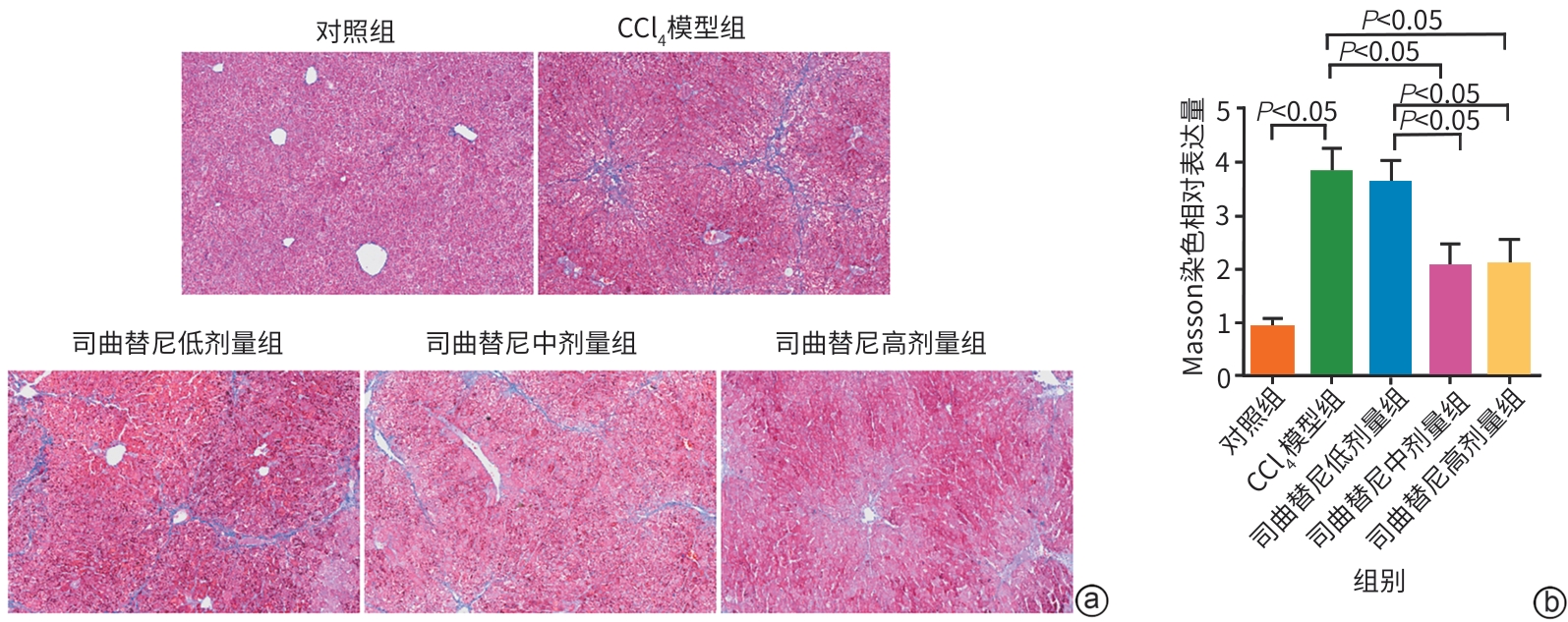
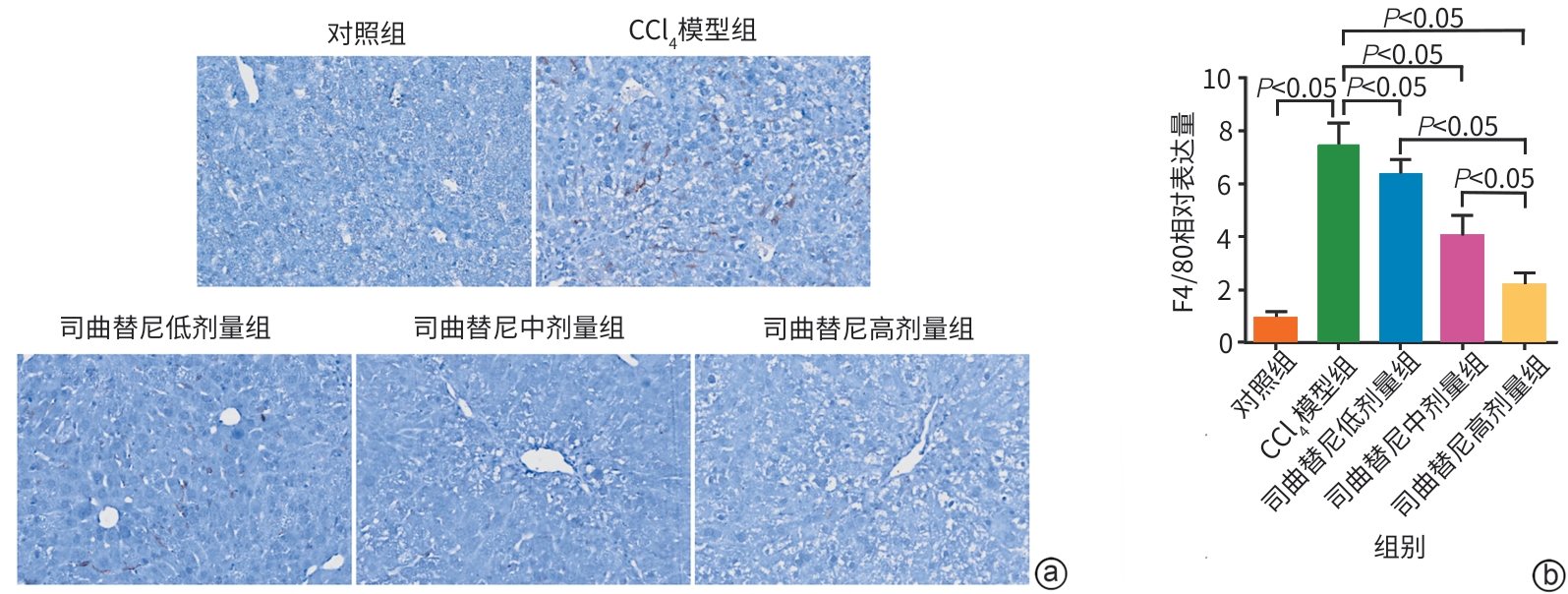
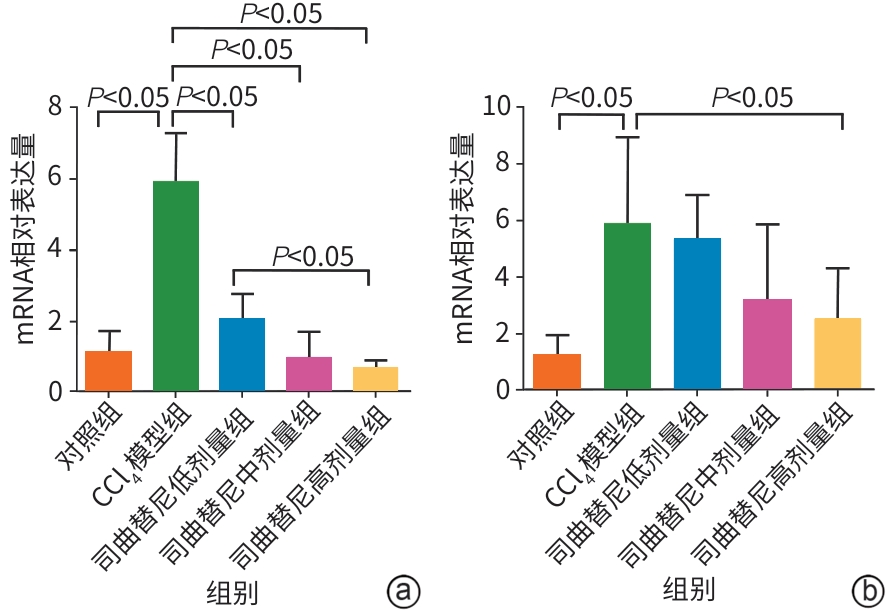
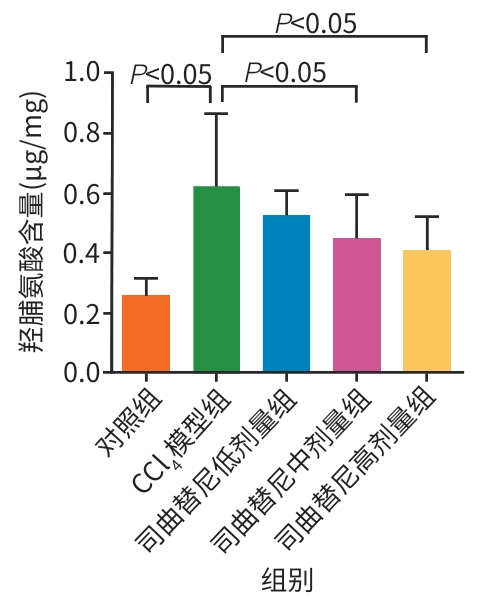
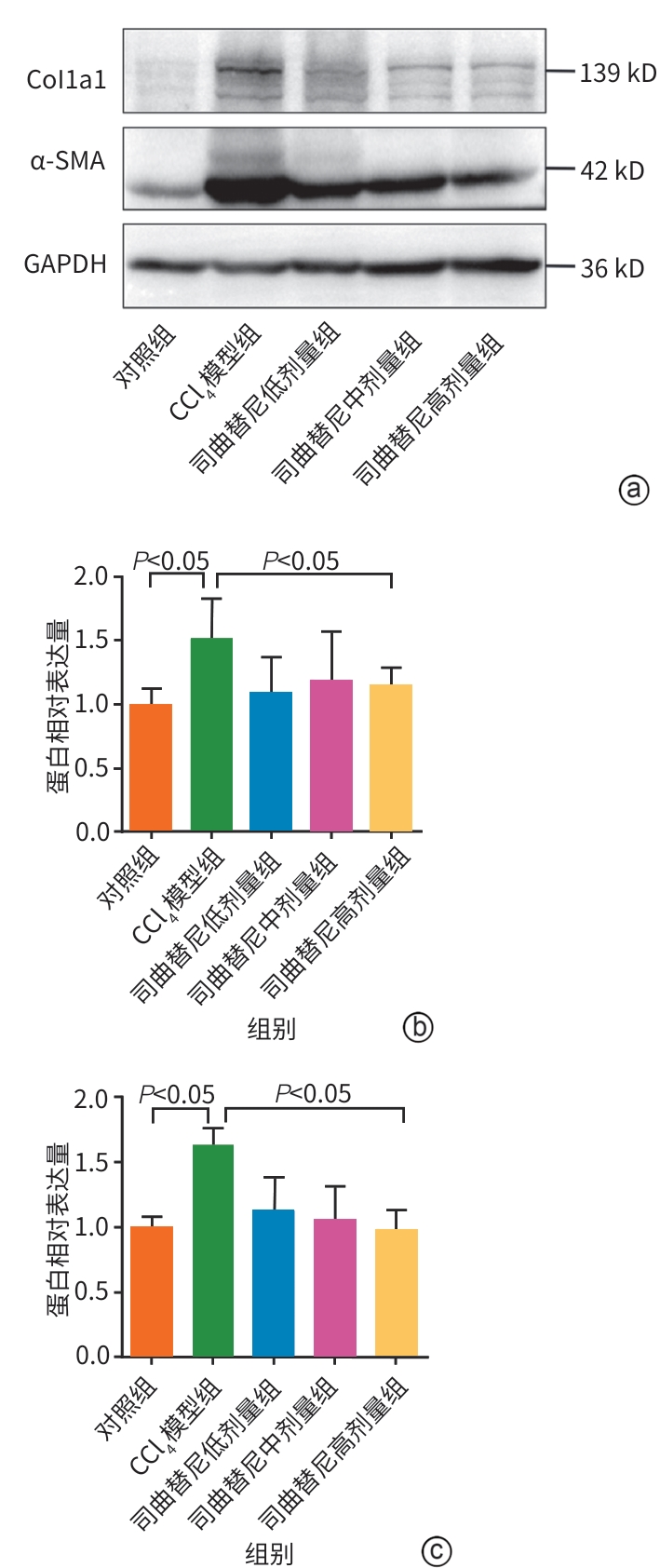
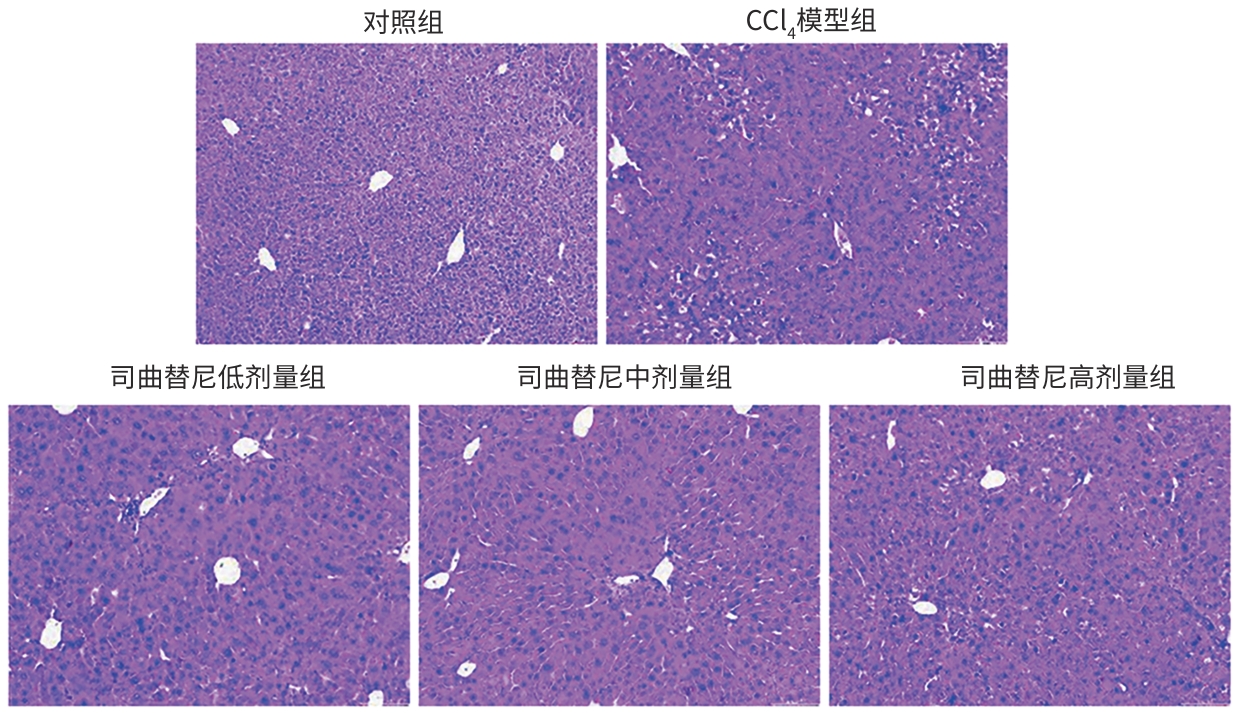
目的 评估司曲替尼对四氯化碳(CCl4)诱导的小鼠肝纤维化的治疗作用。 方法 将30只8周龄雄性C57BL/6J小鼠随机分为对照组、CCl4模型组及司曲替尼低(5 mg/kg)、中(10 mg/kg)、高(20 mg/kg)剂量组。除对照组外,其余各组通过腹腔注射CCl4诱导肝纤维化(持续4周);自造模首日起,各司曲替尼剂量组每日灌胃相应浓度司曲替尼。检测各组小鼠血清总胆固醇(TC)、甘油三酯(TG)、丙氨酸氨基转移酶(ALT)水平;测定肝组织羟脯氨酸含量;通过苏木精-伊红、Masson及天狼星红染色观察肝组织病理改变;采用实时荧光定量聚合酶链反应和Western Blot分别检测肝组织中平滑肌肌动蛋白α与Ⅰ型胶原蛋白α1基因的mRNA及蛋白表达水平,以综合评估药物疗效。计量资料多组间比较采用单因素方差分析,进一步两两比较采用LSD‑t检验。 结果 与对照组相比,模型组TC、TG和ALT水平均显著升高(P值均<0.05);与模型组相比,司曲替尼各剂量组TC、TG和ALT水平差异无统计学意义(P值均>0.05)。经司曲替尼干预后,肝组织羟脯氨酸水平下降,中、高剂量组与CCl4模型组相比,差异均有统计学意义(P值均<0.05)。病理染色结果显示,司曲替尼治疗组胶原沉积减少,纤维间隔变薄、稀疏;司曲替尼高剂量组中,4只小鼠肝纤维化分期为S0~S1,2只为S2~S3,较CCl4模型组肝纤维化程度有所缓解(S3~S4为主)。分子水平检测显示,司曲替尼可下调平滑肌肌动蛋白α与Ⅰ型胶原蛋白α1基因的mRNA及蛋白表达(P值均<0.05)。 结论 司曲替尼能有效缓解CCl4诱导的小鼠肝纤维化,其作用机制可能与抑制肝星状细胞活化及胶原合成有关。 -
关键词:
- 肝纤维化 /
- 司曲替尼 /
- 治疗学 /
- 小鼠, 近交C57BL
Abstract:Objective To investigate the therapeutic effect of sitravatinib on carbon tetrachloride (CCl4)-induced liver fibrosis in mice. Methods A total of 30 male C57BL/6J mice, aged 8 weeks, were randomly divided into control group, CCl4 model group, and low- (5 mg/kg), middle- (10 mg/kg), and high-dose (20 mg/kg) sitravatinib groups. All mice except those in the control group were given intraperitoneal injection of CCl4 for 4 consecutive weeks to induce liver fibrosis, and since the first day of modeling, the mice in the low-, middle-, and high-dose sitravatinib groups were given sitravatinib at the corresponding dose by gavage every day. The serum levels of total cholesterol (TC), triglyceride (TG), and alanine aminotransferase (ALT) were measured for the mice in each group; hepatic hydroxyproline content was measured; HE staining, Masson staining, and Sirius Red staining were used to observe liver histopathological changes; quantitative real-time PCR and Western blot were used to measure the mRNA and protein expression levels of α-smooth muscle actin (α-SMA) and collagen type I alpha 1 (Col1a1) in liver tissue. The therapeutic effect of sitravatinib was assessed based on the above results. A one-way analysis of variance was used for comparison of continuous data between multiple groups, and the least significant difference t-test was used for further comparison between two groups. Results Compared with the control group, the model group had significant increases in the levels of TC, TG, and ALT (all P<0.05), and there were no significant differences in the levels of TC, TG, and ALT between the model group and the low-, middle-, and high-dose sitravatinib groups (all P>0.05). Hepatic hydroxyproline content decreased after sitravatinib intervention, with a significant difference between the middle-/high-dose sitravatinib groups and the CCl4 model group (both P<0.05). Histopathological staining showed that the sitravatinib treatment groups had a reduction in collagen deposition, along with thinning and fragmentation of fibrous septa, and in the high-dose sitravatinib group, 4 mice had a fibrosis stage of S0—S1 and 2 mice had a fibrosis stage of S2—S3, suggesting a certain degree of alleviation of liver fibrosis degree compared with the CCl4 model group (mainly S3—S4). The measurement of related molecules showed that sitravatinib downregulated the mRNA and protein expression levels of α-SMA and Col1a1 (all P<0.05). Conclusion Sitravatinib can effectively alleviate CCl4-induced liver fibrosis in mice, possibly by inhibiting hepatic stellate cell activation and collagen synthesis. -
Key words:
- Hepatic Fibrosis /
- Sitravatinib /
- Therapeutics /
- Mice, Inbred C57BL
-
表 1 PCR引物序列
Table 1. PCR primer sequences
基因 序列 18S RNA 上游引物:5'-CATTCGAACGTCTGCCCTATC-3' 下游引物:5'-CCTGCTGCCTTCCTTGGA-3' ACTA2 上游引物:5'-CCCAGACATCAGGGAGTAATGG-3' 下游引物:5'-TCTATCGGATACTTCAGCGTCA-3' COL1A1 上游引物:5'-CGATGGATTCCCGTTCGAGT-3' 下游引物:5'-AAGGGTGCTGTAGGTGAAGC-3' 注:PCR,聚合酶链反应;ACTA2,平滑肌肌动蛋白α基因;COL1A1,Ⅰ型胶原蛋白α1基因。
表 2 小鼠分组、造模及给药情况
Table 2. Grouping, modeling and drug administration of mice
分组 动物数(只) 给药方案 对照组 6 普通饲料喂养基础上予以玉米油腹腔注射,同时予以等量生理盐水灌胃,连续4周 CCl4模型组 6 普通饲料喂养基础上予以CCl4溶液腹腔注射,等量生理盐水灌胃,1次/1.5 d,连续4周 司曲替尼低剂量组 6 普通饲料喂养基础上予以CCl4溶液腹腔注射,司曲替尼5 mg/kg灌胃,1次/1.5 d,连续4周 司曲替尼中剂量组 6 普通饲料喂养基础上予以CCl4溶液腹腔注射,司曲替尼10 mg/kg灌胃,1次/1.5 d,连续4周 司曲替尼高剂量组 6 普通饲料喂养基础上予以CCl4溶液腹腔注射,司曲替尼20 mg/kg灌胃,1次/1.5 d,连续4周 注:CCl4,四氯化碳。腹腔注射体积分数为10%的CCl4玉米油溶液(CCl4与玉米油的比例为1∶9),注射剂量为5 mL/kg,1次/1.5 d,连续4周。
表 3 各组小鼠血清TG、TC和ALT水平的比较
Table 3. Comparison of serum TG、TC and ALT levels of each group of mice
组别 动物数(只) TG(mg/dL) TC(mg/dL) ALT(U/L) 对照组 6 21.68±7.69 162.10±26.76 63.89±12.50 CCl4模型组 6 100.56±25.901) 243.84±44.731) 254.60±39.891) 司曲替尼低剂量组 6 99.50±16.001) 245.06±32.951) 245.74±74.111) 司曲替尼中剂量组 6 87.80±22.071) 228.33±43.471) 233.62±25.281) 司曲替尼高剂量组 6 84.23±23.711) 222.46±22.971) 224.37±33.621) F值 15.76 5.58 4.83 P值 <0.001 0.002 0.005 注:与对照组比较,1)P<0.05。TG,甘油三酯;TC,总胆固醇;ALT,丙氨酸氨基转移酶;CCl4,四氯化碳。
表 4 各组小鼠肝纤维化分期
Table 4. Liver fibrosis grades of mice in each group
组别 动物数
(只)肝纤维化分期(只) S0 S1 S2 S3 S4 对照组 6 6 0 0 0 0 CCl4模型组 6 0 0 0 3 3 司曲替尼低剂量组 6 0 1 2 2 1 司曲替尼中剂量组 6 0 2 2 1 1 司曲替尼高剂量组 6 1 3 1 1 0 注:CCl4,四氯化碳。
-
[1] DHAR D, BAGLIERI J, KISSELEVA T, et al. Mechanisms of liver fibrosis and its role in liver cancer[J]. Exp Biol Med, 2020, 245( 2): 96- 108. DOI: 10.1177/1535370219898141. [2] HERNANDEZ-GEA V, FRIEDMAN SL. Pathogenesis of liver fibrosis[J]. Annu Rev Pathol, 2011, 6: 425- 456. DOI: 10.1146/annurev-pathol-011110-130246. [3] ZHAO J, HAN M, ZHOU L, et al. TAF and TDF attenuate liver fibrosis through NS5ATP9, TGFβ1/Smad3, and NF-κB/NLRP3 inflammasome signaling pathways[J]. Hepatol Int, 2020, 14( 1): 145- 160. DOI: 10.1007/s12072-019-09997-6. [4] CAI BS, DONGIOVANNI P, COREY KE, et al. Macrophage MerTK promotes liver fibrosis in nonalcoholic steatohepatitis[J]. Cell Metab, 2020, 31( 2): 406- 421. DOI: 10.1016/j.cmet.2019.11.013. [5] CHEN ML, MU XY, LI WG, et al. Effects of α-Arrestin-domain-containing-3 deficiency on PI3K/AKT signaling pathway and proliferation of renal cell carcinoma cells[J]. Prog Mod Biomed, 2020, 20( 13): 2401- 2405. DOI: 10.13241/j.cnki.pmb.2020.13.001.陈暮霖, 穆星宇, 李维国, 等. 敲除含α-Arrestin结构域蛋白3对肾癌细胞PI3K/AKT信号通路及增殖能力的影响[J]. 现代生物医学进展, 2020, 20( 13): 2401- 2405. DOI: 10.13241/j.cnki.pmb.2020.13.001. [6] GONG PP. Axl upregulate autophagy in experimental autoimmune encephalomyelitis via PI3K/Akt/mTOR signaling pathway[D]. Lanzhou: Lanzhou University, 2022. DOI: 10.27204/d.cnki.glzhu.2022.002383.巩盼盼. Axl通过PI3K/Akt/mTOR信号通路上调实验性自身免疫性脑脊髓炎小鼠自噬水平的研究[D]. 兰州: 兰州大学, 2022. DOI: 10.27204/d.cnki.glzhu.2022.002383. [7] ZHAO J, YU XM, HUANG DZ, et al. SAFFRON-103: A phase 1b study of the safety and efficacy of sitravatinib combined with tislelizumab in patients with locally advanced or metastatic non-small cell lung cancer[J]. J Immunother Cancer, 2023, 11( 2): e006055. DOI: 10.1136/jitc-2022-006055. [8] WANG X, PAN HM, CUI JW, et al. SAFFRON-103: A phase Ib study of sitravatinib plus tislelizumab in anti-PD-(L)1 refractory/resistant advanced melanoma[J]. Immunotherapy, 2024, 16( 4): 243- 256. DOI: 10.2217/imt-2023-0130. [9] WANG S, ZHU L, LI TE, et al. Disruption of MerTK increases the efficacy of checkpoint inhibitor by enhancing ferroptosis and immune response in hepatocellular carcinoma[J]. Cell Rep Med, 2024, 5( 2): 101415. DOI: 10.1016/j.xcrm.2024.101415. [10] YANG FM, LI H, LI YM, et al. Crosstalk between hepatic stellate cells and surrounding cells in hepatic fibrosis[J]. Int Immunopharmacol, 2021, 99: 108051. DOI: 10.1016/j.intimp.2021.108051. [11] WANG L, ZHOU JY, WANG J, et al. Hepatic stellate cell-targeting micelle nanomedicine for early diagnosis and treatment of liver fibrosis[J]. Adv Healthc Mater, 2024, 13( 12): e2303710. DOI: 10.1002/adhm.202303710. [12] WANG TT, XIA GQ, LI X, et al. Endoplasmic reticulum stress in liver fibrosis: Mechanisms and therapeutic potential[J]. Biochim Biophys Acta Mol Basis Dis, 2025, 1871( 3): 167695. DOI: 10.1016/j.bbadis.2025.167695. [13] MOHAN CD, SHANMUGAM MK, GOWDA SGS, et al. c-MET pathway in human malignancies and its targeting by natural compounds for cancer therapy[J]. Phytomedicine, 2024, 128: 155379. DOI: 10.1016/j.phymed.2024.155379. [14] RAJ S, KESARI KK, KUMAR A, et al. Molecular mechanism(s) of regulation(s) of c-MET/HGF signaling in head and neck cancer[J]. Mol Cancer, 2022, 21( 1): 31. DOI: 10.1186/s12943-022-01503-1. [15] LUO XX. Experimental study on the dynamic changes of PI3K/Akt signaling pathway in hepatic stellate cells after X-ray irradiation[D]. Urumqi: Xinjiang Medical University, 2016.罗晓旭. PI3K/Akt信号通路与肝星状细胞活化的关系及其在放射性肝纤维化中的作用[D]. 乌鲁木齐: 新疆医科大学, 2016. [16] DAS A, SHERGILL U, THAKUR L, et al. Ephrin B2/EphB4 pathway in hepatic stellate cells stimulates Erk-dependent VEGF production and sinusoidal endothelial cell recruitment[J]. Am J Physiol Gastrointest Liver Physiol, 2010, 298( 6): G908- G915. DOI: 10.1152/ajpgi.00510.2009. -



 PDF下载 ( 25245 KB)
PDF下载 ( 25245 KB)

 下载:
下载: