黄芪甲苷Ⅳ对四氯化碳诱导的肝纤维化小鼠模型的影响及其作用机制
DOI: 10.12449/JCH260316
Effect of astragaloside Ⅳ on a mouse model of carbon tetrachloride-induced liver fibrosis and its mechanism
-
摘要:
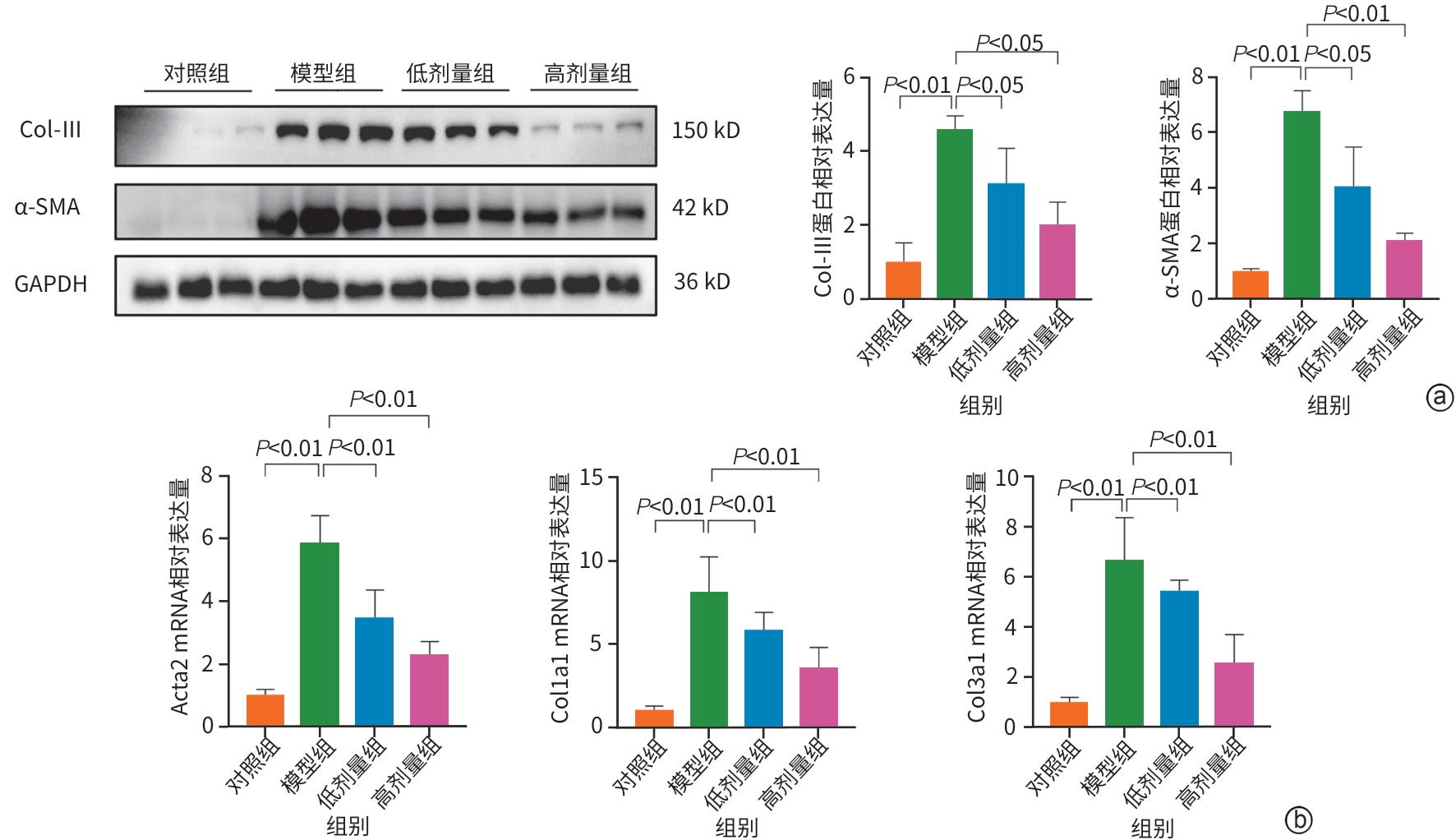
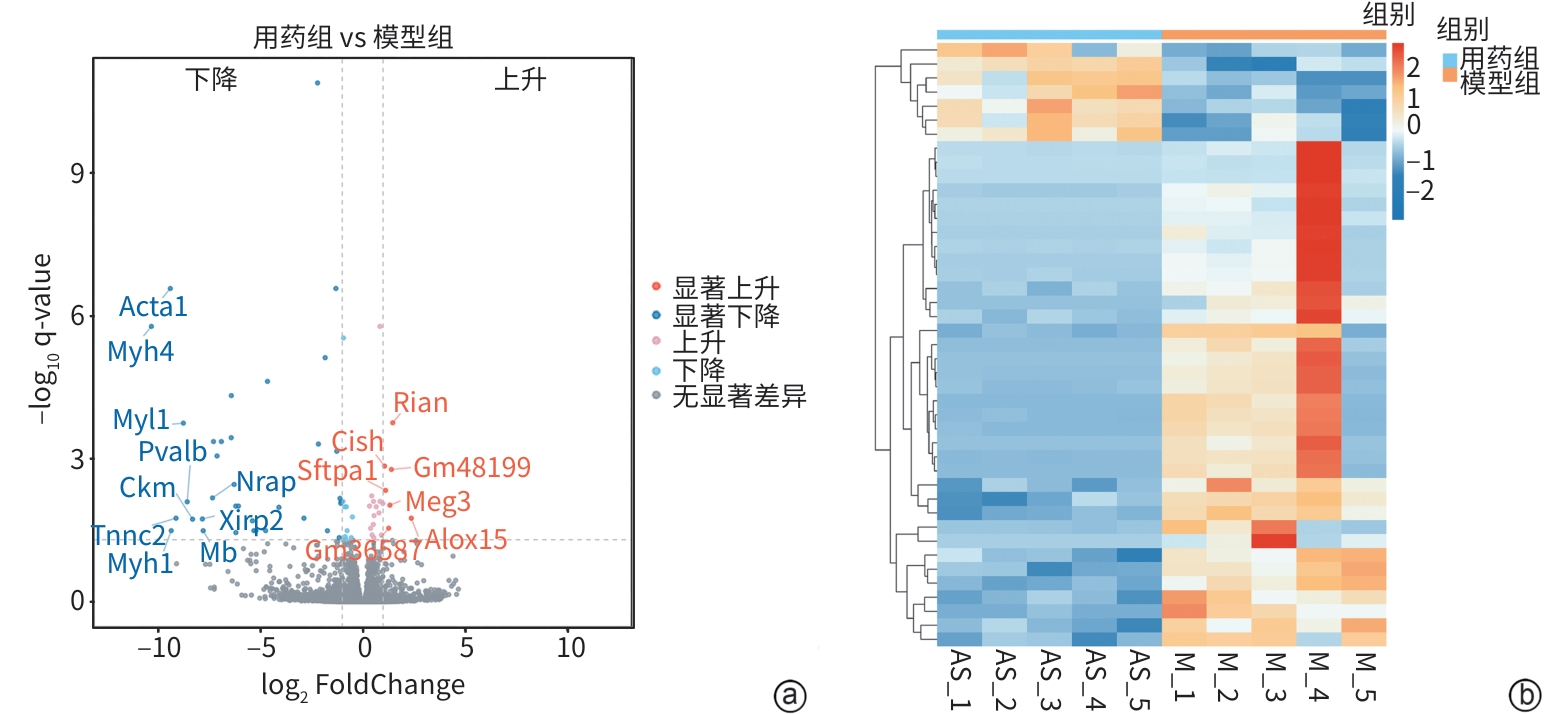
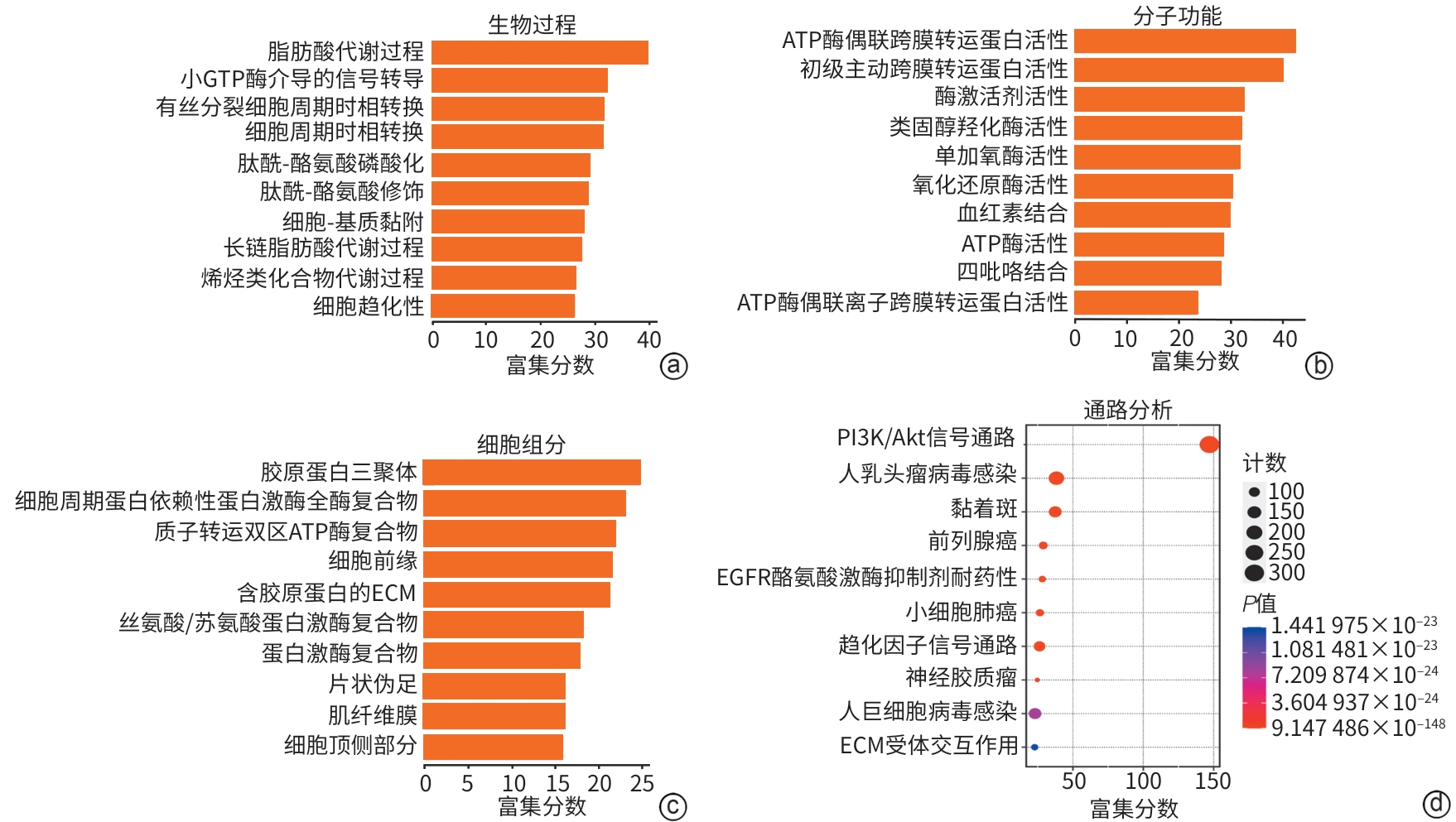
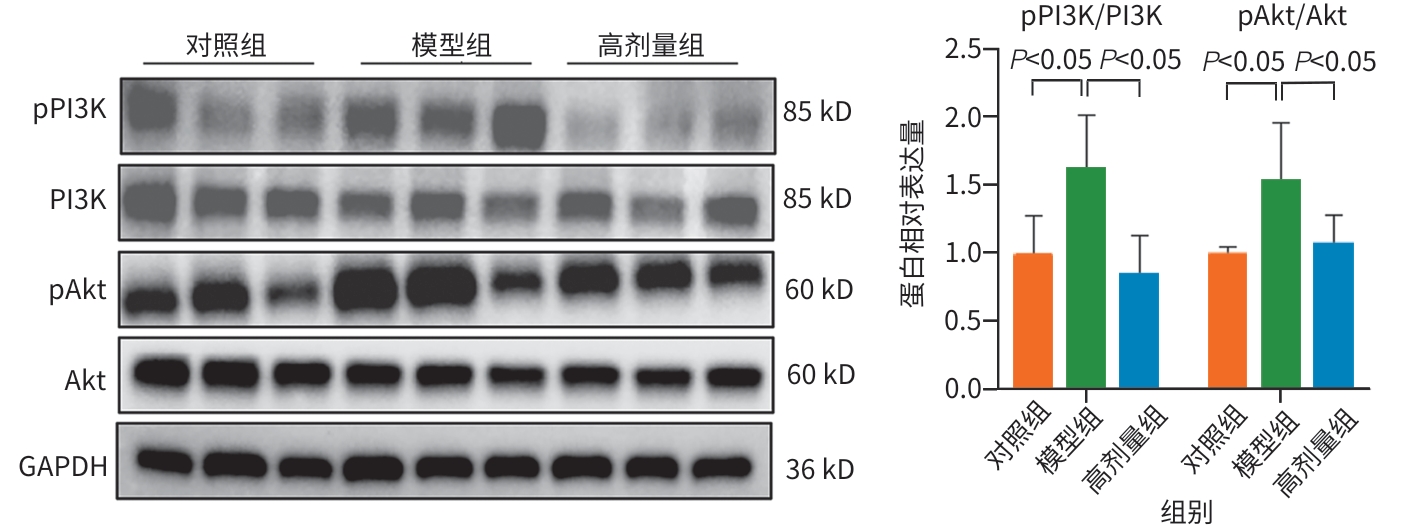
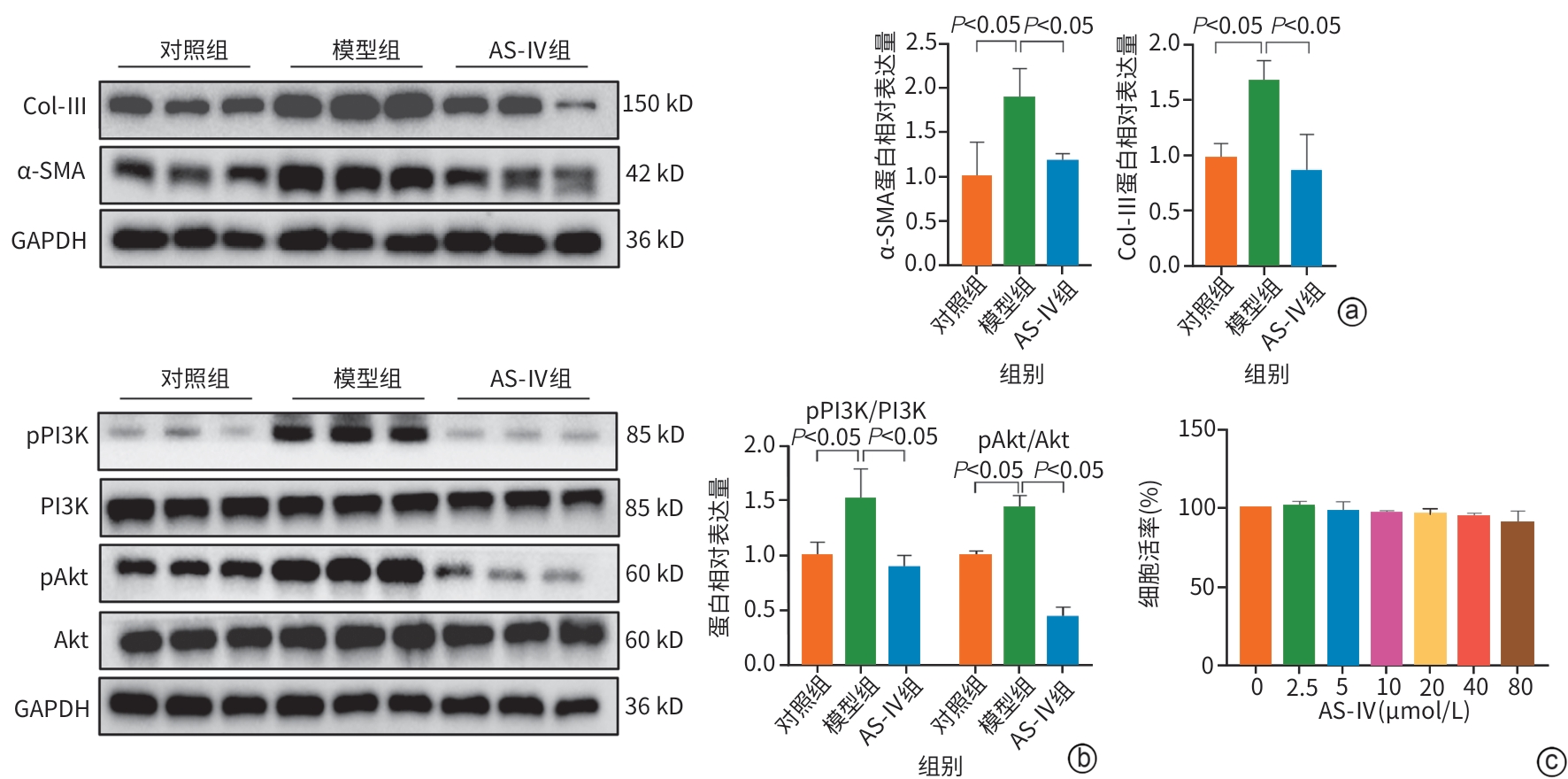
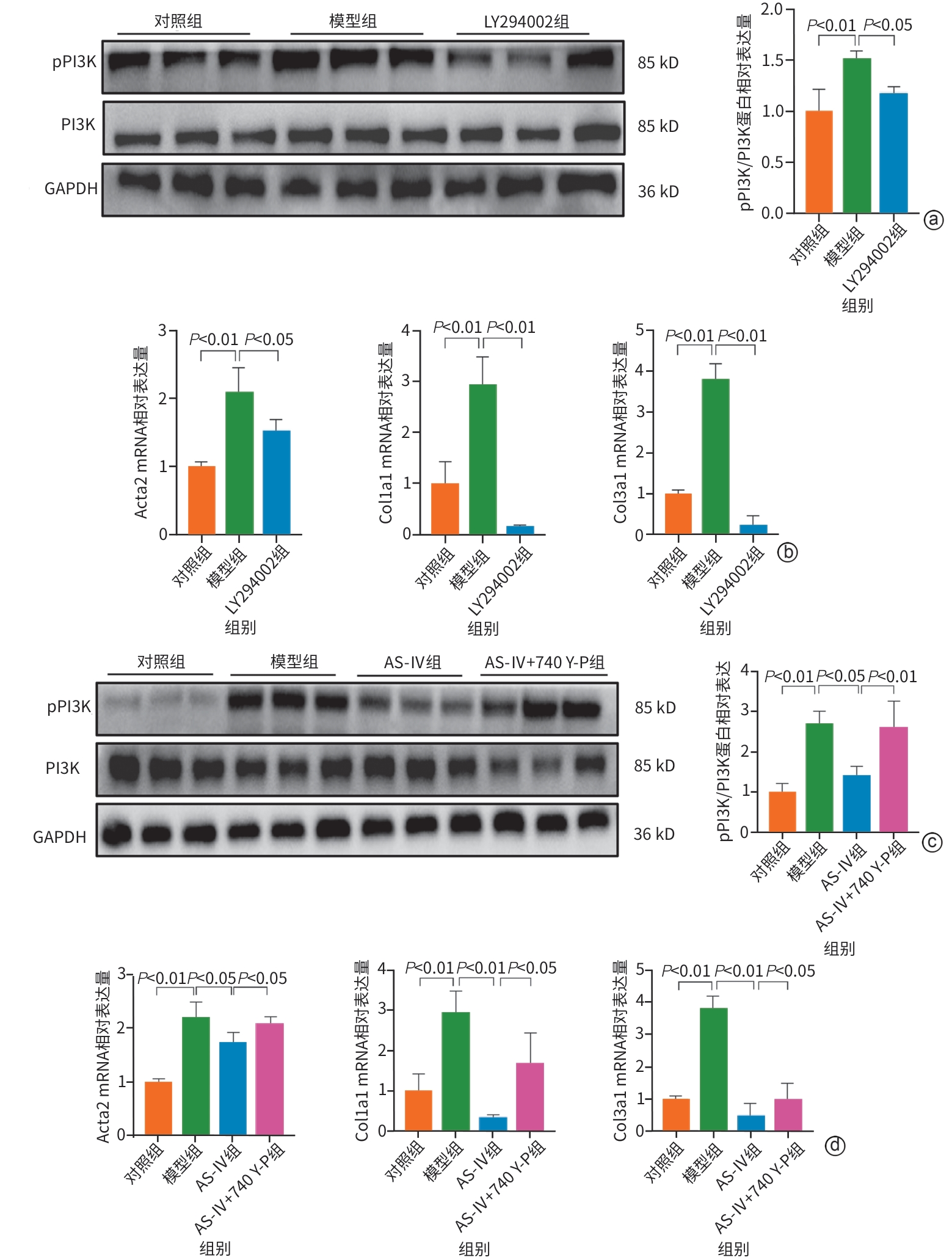
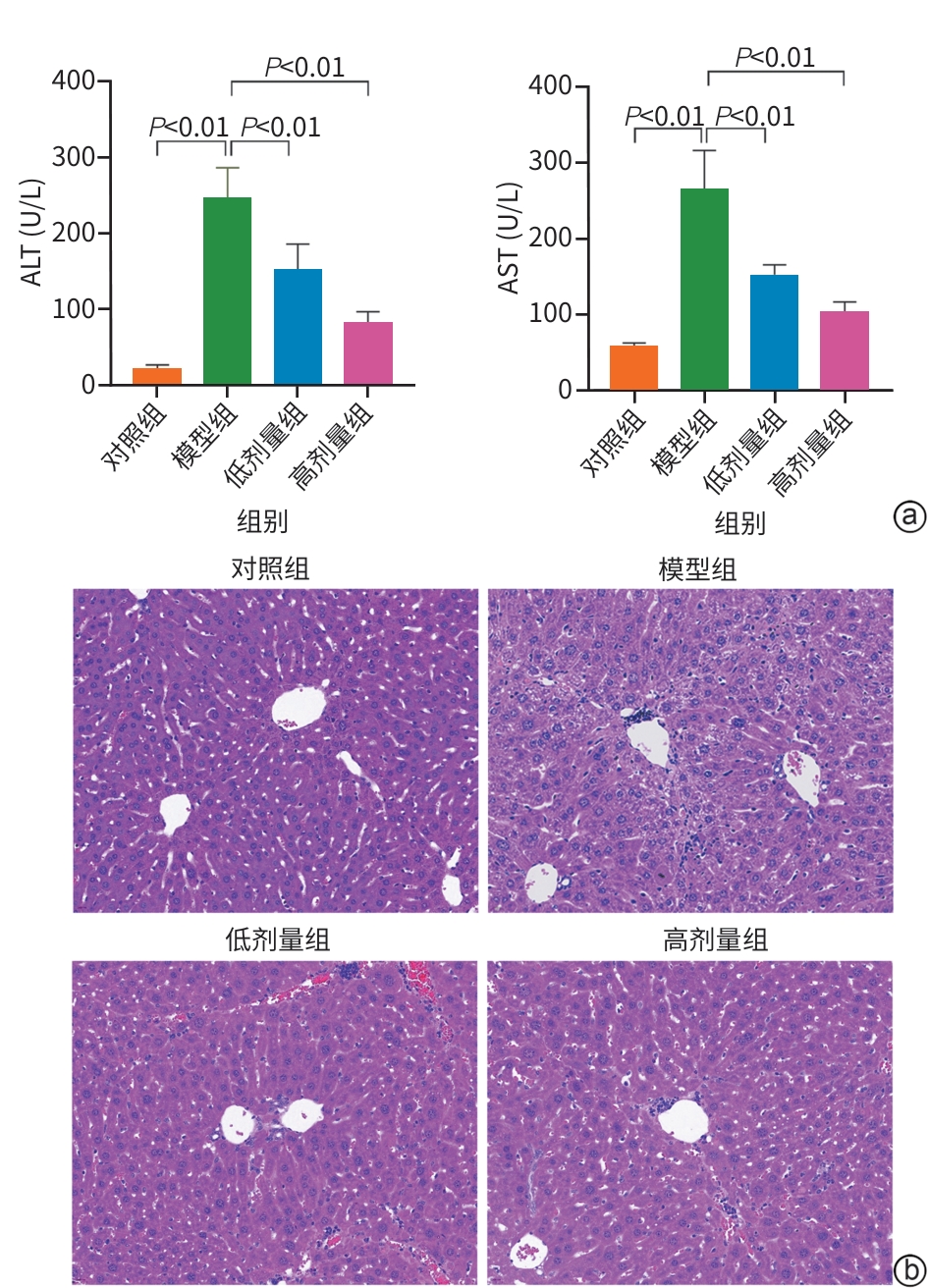
目的 明确黄芪甲苷Ⅳ(AS-Ⅳ)在体内外的肝脏保护和抗肝纤维化作用,并探讨其在抗肝纤维化中的作用机制。 方法 动物实验:将C57BL/6J小鼠分为对照组、模型组、AS-Ⅳ低剂量组(20 mg/kg)和AS-Ⅳ高剂量组(80 mg/kg)。通过腹腔注射四氯化碳6周构建肝纤维化模型,从第3周开始,AS-Ⅳ组分别予以AS-Ⅳ 20 mg/kg、80 mg/kg灌胃,给药4周后检测血清丙氨酸氨基转移酶(ALT)、天冬氨酸氨基转移酶(AST)水平,以及血清透明质酸(HA)、层粘连蛋白(LN)、Ⅲ型前胶原(PⅢNP)及Ⅳ型胶原(Col-Ⅳ)水平;苏木精-伊红染色、天狼星红染色和马松染色观察肝组织病理及胶原沉积情况;定量逆转录聚合酶链反应检测肝组织Acta2、Col1a1、Col3a1的mRNA表达;Western Blot法检测肝组织α-平滑肌肌动蛋白(α-SMA)、Ⅲ型胶原蛋白(Col-Ⅲ)、磷酸化磷脂酰肌醇3-激酶(pPI3K)/PI3K、磷酸化蛋白激酶B(pAkt)/Akt蛋白表达水平;运用转录组学技术对小鼠肝组织进行测序分析,筛选差异基因并进行生物信息学分析。细胞实验:采用转化生长因子β(TGF-β)诱导LX-2细胞活化,进一步使用PI3K抑制剂LY294002以及PI3K激动剂740 Y-P对细胞进行干预,设对照组、模型组、AS-Ⅳ组、LY294002组、AS-Ⅳ+740 Y-P组,36 h后收集细胞。检测LX-2细胞α-SMA、Col-Ⅲ、pPI3K/PI3K和pAkt/Akt蛋白表达的变化,以及Acta2、Col1a1和Col3a1的mRNA相对表达量。计量资料多组间比较采用单因素方差分析,进一步两两比较使用LSD-t检验。 结果 动物实验:与模型组相比,AS-Ⅳ治疗组小鼠血清ALT、AST、HA、LN、PⅢNP和Col-Ⅳ水平均显著降低(P值均<0.01);肝组织Acta2、Col1a1和Col3a1的mRNA表达均显著降低(P值均<0.05);与模型组相比,治疗组小鼠肝组织α-SMA、Col-Ⅲ、pPI3K和pAkt(Ser473)的蛋白表达均显著减少(P值均<0.05)。细胞实验:与对照组相比,TGF-β诱导后模型组细胞α-SMA、Col-Ⅲ、pPI3K和pAkt蛋白表达水平显著升高(P值均<0.05);与模型组比较,AS-Ⅳ组细胞α-SMA、Col-Ⅲ、pPI3K和pAkt(Ser473)蛋白表达水平显著降低(P值均<0.05),LY294002组细胞pPI3K蛋白表达,以及Acta2、Col1a1和Col3a1的mRNA相对表达量亦显著降低(P值均<0.05)。而予740 Y-P干预后,与AS-Ⅳ组相比,pPI3K蛋白表达及Acta2、Col1a1和Col3a1的mRNA相对表达量均明显升高(P值均<0.05)。 结论 AS-Ⅳ通过抑制肝星状细胞活化从而改善肝纤维化,其作用机制可能与抑制PI3K/Akt信号通路有关。 -
关键词:
- 肝纤维化 /
- 黄芪甲苷Ⅳ /
- 小鼠, 近交C57BL /
- 肝星状细胞
Abstract:Objective To investigate the liver-protecting and anti-liver fibrosis effects of astragaloside Ⅳ (AS-Ⅳ) in vitro and in vivo, as well as its mechanism of action in intervention against liver fibrosis. Methods In the animal experiment, C57BL/6J mice were divided into control group, model group, low-dose AS-Ⅳ (20 mg/kg) group, and high-dose AS-Ⅳ (80 mg/kg) group. The mice were given intraperitoneal injection of carbon tetrachloride for 6 weeks to induce liver fibrosis, and since week 3 of injection, the mice in the low-dose AS-Ⅳ group and the high-dose AS-Ⅳ group were given AS-Ⅳ by gavage at a dose of 20 mg/kg and 80 mg/kg, respectively. The serum levels of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were measured after 4 weeks of administration, as well as the serum levels of hyaluronic acid (HA), laminin (LN), procollagen Ⅲ N-terminal peptide (PⅢNP), and collagen type Ⅳ (Col-Ⅳ). HE staining, picrosirius red staining, and Masson staining were used to observe liver histopathology and collagen deposition; RT-qPCR was used to measure the mRNA expression levels of Acta2, Col1a1, and Col3a1 in liver tissue, and Western blot was used to measure the protein expression levels of α-smooth muscle actin (α-SMA), collagen type Ⅲ (Col-Ⅲ), phosphatidylinositol 3-kinase (PI3K), phosphorylated PI3K (pPI3K), protein kinase B (Akt), and phosphorylated AKT (p-Akt) in liver tissue; transcriptome sequencing was performed for liver tissue to identify differentially expressed genes and perform a bioinformatics analysis. In the cell experiment, transforming growth factor-β (TGF-β) was used to induce the activation of LX-2 cells, and the PI3K inhibitor LY294002 and the PI3K activator 740 Y-P were used for intervention. The cells were divided into control group, model group, AS-Ⅳ group, LY294002 group, and AS-Ⅳ+740 Y-P group, and the cells were harvested after 36 hours of intervention. Changes in the protein expression levels of α-SMA, Col-Ⅲ, pPI3K/PI3K, and pAkt/Akt in LX-2 cells were measured, as well as changes in the relative mRNA expression levels of Acta2, Col1a1, and Col3a1. A one-way analysis of variance was used for comparison of continuous data between multiple groups, and the least significant difference t-test was used for further comparison between two groups. Results In the animal experiment, compared with the model group, the AS-Ⅳ treatment group had significant reductions in the serum levels of ALT, AST, HA, LN, PⅢNP, and Col-Ⅳ (all P<0.01), the mRNA expression levels of Acta2, Col1a1, and Col3a1 in liver tissue (all P<0.05), and the protein expression levels of α-SMA, Col-Ⅲ, pPI3K, and pAkt (Ser473) in liver tissue (all P<0.05). In the cell experiment, compared with the control group, the model group had significant increases in the protein expression levels of α-SMA, Col-Ⅲ, pPI3K, and pAkt (Ser473) after TGF-β induction (all P<0.05); compared with the model group, the AS-Ⅳ group had significant reductions in the protein expression levels of α-SMA, Col-Ⅲ, pPI3K, and pAkt (Ser473) (all P<0.05), and both the AS-Ⅳ group and the LY294002 group had significant reductions in the protein expression level of pPI3K and the relative mRNA expression levels of Acta2, Col1a1, and Col3a1 (all P<0.05). Compared with the AS-Ⅳ group, there were significant increases in the protein expression level of pPI3K and the relative mRNA expression levels of Acta2, col1a1, and Col3a1 after 740 Y-P intervention (all P<0.05). Conclusion AS-Ⅳ can inhibit hepatic stellate cell activation and improve liver fibrosis, possibly by inhibiting the PI3K/Akt signaling pathway. -
Key words:
- Hepatic Fibrosis /
- Astragaloside Ⅳ /
- Mice, Inbred C57BL /
- Hepatic Stellate Cells
-
表 1 引物序列
Table 1. Primer sequences
基因 正向(5'-3') 反向(5'-3') Col3a1 小鼠肝组织 CTGTAACATGGAAACTGGGGAAA CCATAGCTGAACTGAAAACCACC 人LX-2细胞 CGTGGTAGCCCTGGTGAGAGAG TGGAGAACCGCTGGGACCTG Col1a1 小鼠肝组织 CAGGCTGGTGTGATGGGATT CGTTCTCCGCTCTCTCCAAA 人LX-2细胞 GAGGGCCAAGACGAAGACATC CAGATCACGTCATCGCACAAC Acta2 小鼠肝组织 TATCCCCGGGACTAAGACGG CTGTAGTCCCCCACTACCAC 人LX-2细胞 CTCTGGACGCACAACTGGCATC CCCATCAGGCAACTCGTAACTCTTC GAPDH 小鼠肝组织 AGGTCGGTGTGAACGGATTTG TGTAGACCATGTAGTTGAGGTCA 人LX-2细胞 GGAGCGAGATCCCTCCAAAAT GGCTGTTGTCATACTTCTCATGG -
[1] HUANG DQ, TERRAULT NA, TACKE F, et al. Global epidemiology of cirrhosis: Aetiology, trends and predictions[J]. Nat Rev Gastroenterol Hepatol, 2023, 20( 6): 388- 398. DOI: 10.1038/s41575-023-00759-2. [2] ZHANG CY, LIU S, YANG M. Treatment of liver fibrosis: Past, current, and future[J]. World J Hepatol, 2023, 15( 6): 755- 774. DOI: 10.4254/wjh.v15.i6.755. [3] TRIVEDI P, WANG S, FRIEDMAN SL. The power of plasticity-metabolic regulation of hepatic stellate cells[J]. Cell Metab, 2021, 33( 2): 242- 257. DOI: 10.1016/j.cmet.2020.10.026. [4] YIN CY, EVASON KJ, ASAHINA K, et al. Hepatic stellate cells in liver development, regeneration, and cancer[J]. J Clin Invest, 2013, 123( 5): 1902- 1910. DOI: 10.1172/JCI66369. [5] ZHANG JQ, WU CX, GAO L, et al. Astragaloside IV derived from Astragalus membranaceus: A research review on the pharmacological effects[J]. Adv Pharmacol, 2020, 87: 89- 112. DOI: 10.1016/bs.apha.2019.08.002. [6] HUANG SW, SHAO XH, ZHANG X, et al. Effects of astragaloside IV on low-glucose mediated tumor immunosuppression microenvironment and its mechanism[J]. Chin J Clin Pharmacol Ther, 2024, 29( 11): 1201- 1211. DOI: 10.12092/j.issn.1009-2501.2024.11.001.黄仕文, 邵晓寒, 张雪, 等. 黄芪甲苷Ⅳ干预低糖介导的肿瘤免疫抑制微环境作用及其机制研究[J]. 中国临床药理学与治疗学, 2024, 29( 11): 1201- 1211. DOI: 10.12092/j.issn.1009-2501.2024.11.001. [7] LIANG XY, HONG FF, YANG SL. Astragaloside IV alleviates liver inflammation, oxidative stress and apoptosis to protect against experimental non-alcoholic fatty liver disease[J]. Diabetes Metab Syndr Obes, 2021, 14: 1871- 1883. DOI: 10.2147/DMSO.S304817. [8] ZHANG C, LI LL, HOU S, et al. Astragaloside IV inhibits hepatocellular carcinoma by continually suppressing the development of fibrosis and regulating pSmad3C/3L and Nrf2/HO-1 pathways[J]. J Ethnopharmacol, 2021, 279: 114350. DOI: 10.1016/j.jep.2021.114350. [9] CHEN HX, CAI JY, WANG JC, et al. Targeting Nestin+ hepatic stellate cells ameliorates liver fibrosis by facilitating TβRI degradation[J]. J Hepatol, 2021, 74( 5): 1176- 1187. DOI: 10.1016/j.jhep.2020.11.016. [10] YAN MY, CUI YM, XIANG Q. Metabolism of hepatic stellate cells in chronic liver diseases: Emerging molecular and therapeutic interventions[J]. Theranostics, 2025, 15( 5): 1715- 1740. DOI: 10.7150/thno.106597. [11] SUN YX, YUAN XY, HU ZH, et al. Harnessing nuclear receptors to modulate hepatic stellate cell activation for liver fibrosis resolution[J]. Biochem Pharmacol, 2025, 232: 116730. DOI: 10.1016/j.bcp.2024.116730. [12] HUANG FJ, XIE XX, LIU X, et al. Research progress on the active ingredients of traditional Chinese medicine against hepatic fibrosis by regulating the MAPK signaling pathway[J]. Chin J Hosp Pharm, 2025, 45( 2): 225- 233. DOI: 10.13286/j.1001-5213.2025.02.17.黄甫静, 谢星星, 刘学, 等. 中药活性成分调控MAPK信号通路抗肝纤维化的研究进展[J]. 中国医院药学杂志, 2025, 45( 2): 225- 233. DOI: 10.13286/j.1001-5213.2025.02.17. [13] CHEN SP, KANG RD, HONG XB, et al. Astragaloside IV regulates Snail1 lactylation and acetylation to mediate macrophage polarization and improve myocardial infarction[J]. Chin J Cell Mol Immunol, 2025, 41( 4): 289- 299. DOI: 10.13423/j.cnki.cjcmi.009952.陈少鹏, 康儒典, 洪新宝, 等. 黄芪甲苷Ⅳ调控Snail1乳酸化和乙酰化介导巨噬细胞极化改善大鼠心肌梗死[J]. 细胞与分子免疫学杂志, 2025, 41( 4): 289- 299. DOI: 10.13423/j.cnki.cjcmi.009952. [14] LI HL, WANG P, HUANG F, et al. Astragaloside IV protects blood-brain barrier integrity from LPS-induced disruption via activating Nrf2 antioxidant signaling pathway in mice[J]. Toxicol Appl Pharmacol, 2018, 340: 58- 66. DOI: 10.1016/j.taap.2017.12.019. [15] QING LY, LIU LT, XU QP, et al. Astragaloside IV attenuates pathological myocardial hypertrophy and fibrosis in mice via EGR1-SIRT1-PPARα-SCAD signaling pathway[J]. Chin Pharmacol Bull, 2025, 41( 2): 242- 250. DOI: 10.12360/CPB202406092.卿丽媛, 刘兰婷, 徐庆萍, 等. 黄芪甲苷Ⅳ基于EGR1-SIRT1-PPARα-SCAD信号通路减轻小鼠病理性心肌肥厚及纤维化[J]. 中国药理学通报, 2025, 41( 2): 242- 250. DOI: 10.12360/CPB202406092. [16] SHAMSAN E, ALMEZGAGI M, GAMAH M, et al. The role of PI3k/AKT signaling pathway in attenuating liver fibrosis: A comprehensive review[J]. Front Med, 2024, 11: 1389329. DOI: 10.3389/fmed.2024.1389329. [17] WU C, HUANG L, MO LQ, et al. Anti-fibrotic effects of salvianolate on hepatic fibrosis in rats by regulating TGF-β1/Smad and PI3K/AKT/mTOR signaling pathway[J]. Chin J Hosp Pharm, 2019, 39( 7): 670- 675. DOI: 10.13286/j.cnki.chinhosppharmacyj.2019.07.04.吴灿, 黄亮, 莫立乾, 等. 丹参多酚酸盐通过TGF-β1/Smad和PI3K/AKT/mTOR信号通路抑制大鼠肝纤维化的进展[J]. 中国医院药学杂志, 2019, 39( 7): 670- 675. DOI: 10.13286/j.cnki.chinhosppharmacyj.2019.07.04. [18] WEI LW, CHEN QS, GUO AJ, et al. Asiatic acid attenuates CCl4-induced liver fibrosis in rats by regulating the PI3K/AKT/mTOR and Bcl-2/Bax signaling pathways[J]. Int Immunopharmacol, 2018, 60: 1- 8. DOI: 10.1016/j.intimp.2018.04.016. [19] AKKıZ H, GIESELER RK, CANBAY A. Liver fibrosis: From basic science towards clinical progress, focusing on the central role of hepatic stellate cells[J]. Int J Mol Sci, 2024, 25( 14): 7873. DOI: 10.3390/ijms25147873. [20] BAI XY, ZHANG X, HAI L, et al. Regulatory effect and mechanism of macrophage polarization in liver fibrosis[J]. J Clin Hepatol, 2024, 40( 3): 611- 615. DOI: 10.12449/JCH240329.白小洋, 张旭, 海龙, 等. 巨噬细胞极化在肝纤维化中的调控作用机制[J]. 临床肝胆病杂志, 2024, 40( 3): 611- 615. DOI: 10.12449/JCH240329. [21] GAO YY, ZHAN XY, ZHOU HM. The role of endoplasmic reticulum stress in regulating macrophage immune response in liver diseases[J]. Organ Transpl, 2024, 15( 6): 889- 894. DOI: 10.3969/j.issn.1674-7445.2024144.高逸云, 詹欣雨, 周浩明. 内质网应激调控巨噬细胞免疫应答在肝脏疾病中的作用[J]. 器官移植, 2024, 15( 6): 889- 894. DOI: 10.3969/j.issn.1674-7445.2024144. [22] WANG LP, HUANG Y, CHEN JR, et al. Dynamic crosstalk between HSCs and liver microenvironment: Multicellular interactions in the regulation of liver fibrosis[J]. Front Cell Dev Biol, 2025, 13: 1635763. DOI: 10.3389/fcell.2025.1635763. [23] DUFFIELD JS, FORBES SJ, CONSTANDINOU CM, et al. Selective depletion of macrophages reveals distinct, opposing roles during liver injury and repair[J]. J Clin Invest, 2005, 115( 1): 56- 65. DOI: 10.1172/JCI22675. [24] FENG M, DING J, WANG M, et al. Kupffer-derived matrix metalloproteinase-9 contributes to liver fibrosis resolution[J]. Int J Biol Sci, 2018, 14( 9): 1033- 1040. DOI: 10.7150/ijbs.25589. [25] NI J, HONG XD, JI K, et al. Influence of hepatitis B combined with hepatic fibrosis on endogenous and exogenous metabolism and therapeutic implications[J]. Chin J Clin Pharmacol Ther, 2024, 29( 8): 853- 860. DOI: 10.12092/j.issn.1009-2501.2024.08.002.倪姐, 洪小丹, 计可, 等. 乙肝合并肝纤维化对内外源性代谢的影响及治疗学启示[J]. 中国临床药理学与治疗学, 2024, 29( 8): 853- 860. DOI: 10.12092/j.issn.1009-2501.2024.08.002. [26] SUGIMOTO A, SAITO Y, WANG GX, et al. Hepatic stellate cells control liver zonation, size and functions via R-spondin 3[J]. Nature, 2025, 640( 8059): 752- 761. DOI: 10.1038/s41586-025-08677-w. -



 PDF下载 ( 30584 KB)
PDF下载 ( 30584 KB)

 下载:
下载: