肝内巨噬细胞在代谢相关脂肪性肝病中的调控作用及其机制
DOI: 10.12449/JCH260323
Regulatory role and mechanism of intrahepatic macrophages in metabolic dysfunction-associated fatty liver disease
-
摘要: 随着生活水平提高,代谢相关脂肪性肝病(MAFLD)的发病率日趋上升,且呈现年轻化趋势,已成为全球性健康问题,然而其具体发病机制尚未明确。巨噬细胞作为参与MAFLD发病的重要细胞之一,具有抗原提呈、清除病原微生物以及促进肝脏炎症反应等重要功能,长期以来备受关注。最新研究表明,巨噬细胞或将成为MAFLD的新型治疗靶点。本文系统综述了肝内巨噬细胞在MAFLD相关肝脏炎症中的作用,包括其活化、极化、募集机制以及与其他细胞的相互作用,旨在为MAFLD的临床预防和治疗提供新思路与视角。Abstract: With the improvement of living standards, the incidence rate of metabolic dysfunction-associated fatty liver disease (MAFLD) is gradually increasing with a younger age of onset, and MAFLD has become a global health problem, while its specific pathogenesis remains unclear. Macrophages, as one of the important cells involved in the pathogenesis of MAFLD, have the ability to present antigens, eliminate pathogenic microorganisms, and promote liver inflammatory responses, thereby attracting wide attention for a long time. The latest studies have shown that macrophages may become a new therapeutic target for MAFLD. This article systematically reviews the role of intrahepatic macrophages in liver inflammation caused by MAFLD, including their activation, polarization, recruitment mechanisms, and interactions with other cells, in order to provide new ideas and perspectives for the clinical prevention and treatment of MAFLD.
-
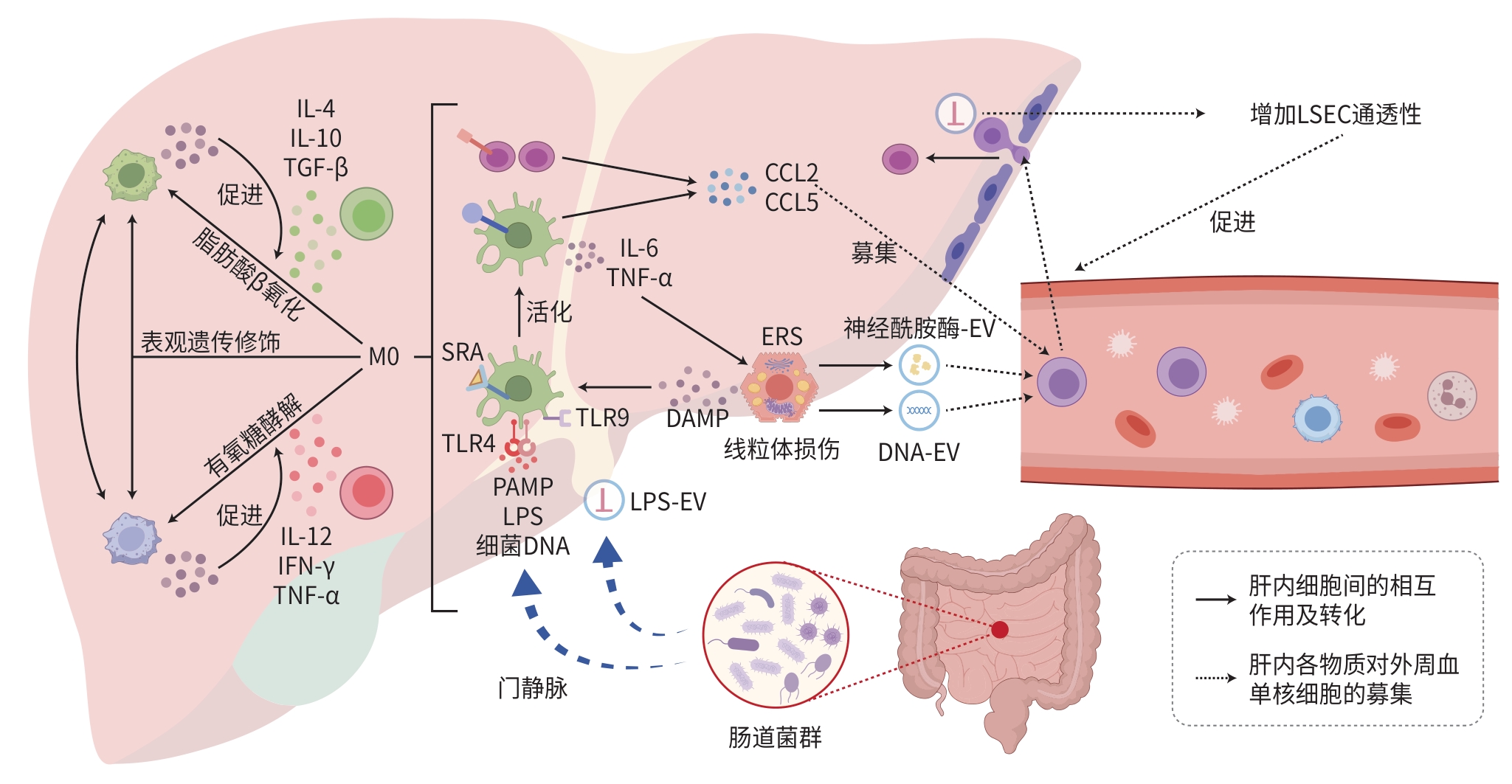
注: MAFLD,代谢相关脂肪性肝病;IL-4,白细胞介素4;IL-10,白细胞介素-10;TGF-β,转化生长因子β;IL-12,白细胞介素12;IFN-γ,干扰素γ;TNF-α,肿瘤坏死因子α;IL-6,白细胞介素6;LSEC,肝窦内皮细胞;M0,静息状态的巨噬细胞;DAMP,损伤相关分子模式;PAMP,病原体相关分子模式;LPS,脂多糖;TLR,Toll样受体;SRA,清道夫受体A;ERS,内质网应激;EV,细胞外囊泡;CCL2,CC亚族趋化因子配体2;CCL5,CC亚族趋化因子配体5。
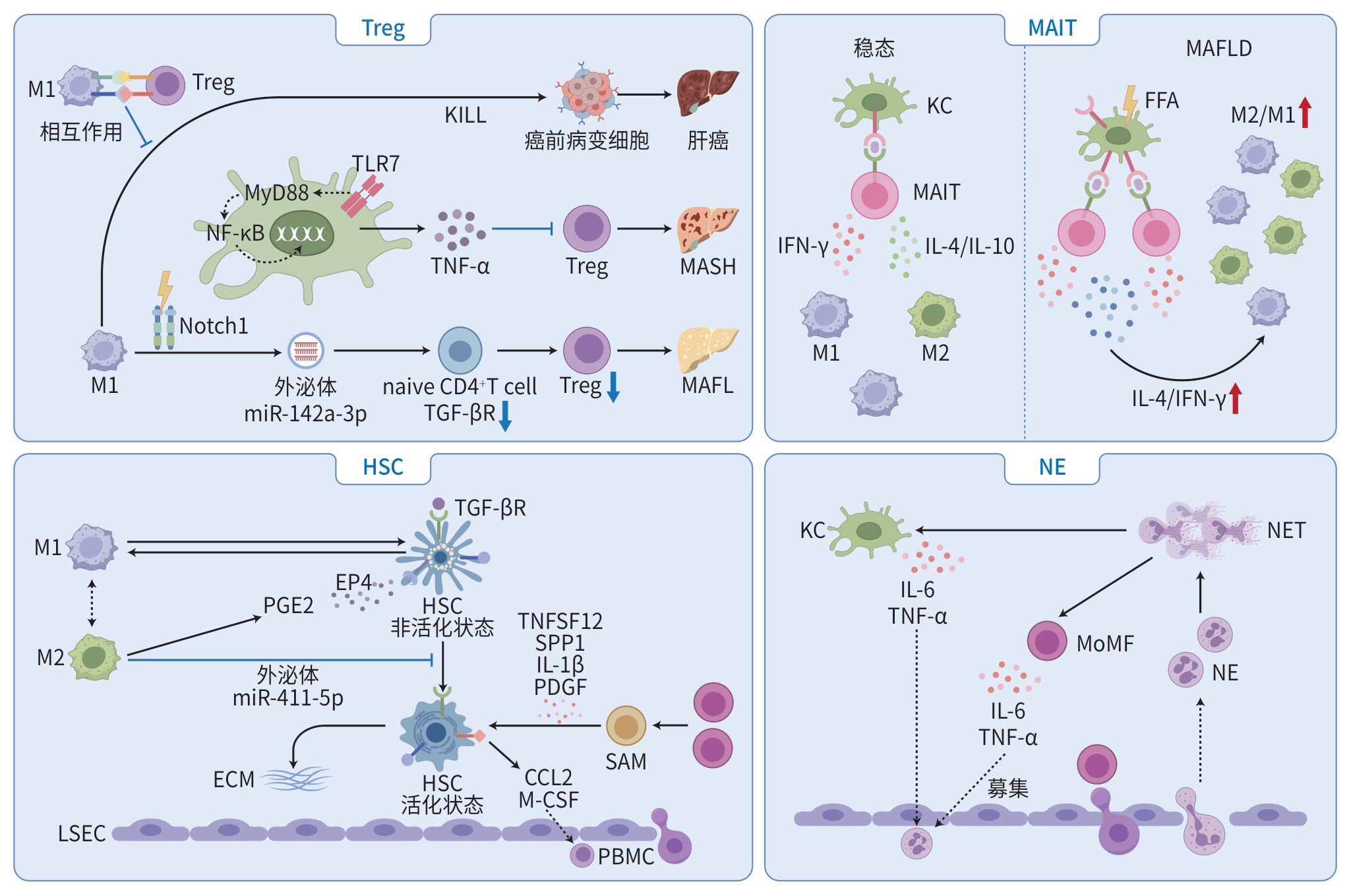
图 1 肝内巨噬细胞在MAFLD发生发展中的活化、极化、募集机制
Figure 1. The mechanisms of activation, polarization, recruitment of intrahepatic macrophages in the development of MAFLD
注: HSC,肝星状细胞;Treg,调节性T细胞;NE,中性粒细胞;NET,中性粒细胞外诱捕网;SAM,瘢痕相关巨噬细胞;TNFSF12,肿瘤坏死因子超家族成员12;M-CSF,巨噬细胞集落刺激因子;PDGF,血小板源性生长因子;SPP1,分泌性磷蛋白1;PGE2,前列腺素E2;ECM,细胞外基质;TLR7,Toll样受体7;MyD88,髓系分化初级反应蛋白质88;NF-κB,核因子κB;miR-142a-3p,微RNA-142a-3p;TGF-βR,转化生长因子β受体;TNF-α,肿瘤坏死因子α;MAFL,代谢相关性脂肪肝;MASH,代谢相关脂肪性肝炎;MAFLD,代谢相关脂肪性肝病;KC,肝巨噬细胞;MoMF,单核细胞来源巨噬细胞;PBMC,外周血单核细胞;MAIT,黏膜相关恒定T细胞;LSEC,肝窦内皮细胞;CCL2,CC亚族趋化因子配体2;IL-6,白细胞介素6;IFNγ,干扰素γ;EP4,前列腺素E2受体4;FFA,游离脂肪酸。
图 2 肝内巨噬细胞在MAFLD发生发展中与其他细胞的相互作用
Figure 2. The schematic diagram of the interaction between intrahepatic macrophages and other cells in the development of MAFLD
-
[1] TARGHER G, BYRNE CD, TILG H. MASLD: A systemic metabolic disorder with cardiovascular and malignant complications[J]. Gut, 2024, 73( 4): 691- 702. DOI: 10.1136/gutjnl-2023-330595. [2] JIANG J, GAO YQ, WANG J, et al. Hepatic sphingomyelin phosphodiesterase 3 promotes steatohepatitis by disrupting membrane sphingolipid metabolism[J]. Cell Metab, 2025, 37( 5): 1119- 1136.e13. DOI: 10.1016/j.cmet.2025.01.016. [3] BARREBY E, CHEN P, AOUADI M. Macrophage functional diversity in NAFLD: More than inflammation[J]. Nat Rev Endocrinol, 2022, 18( 8): 461- 472. DOI: 10.1038/s41574-022-00675-6. [4] KRENKEL O, HUNDERTMARK J, ABDALLAH AT, et al. Myeloid cells in liver and bone marrow acquire a functionally distinct inflammatory phenotype during obesity-related steatohepatitis[J]. Gut, 2020, 69( 3): 551- 563. DOI: 10.1136/gutjnl-2019-318382. [5] WANG NX, LI YR, CAO ZY, et al. Dynamic regulatory mechanisms of macrophage subpopulations in MASH[J/OL]. Chin J Immunol, 2025.( 2025-06-06). https://kns.cnki.net/kcms/detail/22.1126.r.20250606.0937.002.html. https://link.cnki.net/urlid/22.1126.r.20250606.0937.002.html王宁馨, 李怡然, 曹卓妍, 等. 巨噬细胞亚群在MASH进展中的动态调控机制[J/OL]. 中国免疫学杂志, 2025.( 2025-06-06). https://link.cnki.net/urlid/22.1126.r.20250606.0937.002.html. https://link.cnki.net/urlid/22.1126.r.20250606.0937.002.html [6] XIONG XL, KUANG H, ANSARI S, et al. Landscape of intercellular crosstalk in healthy and NASH liver revealed by single-cell secretome gene analysis[J]. Mol Cell, 2019, 75( 3): 644- 660.e5. DOI: 10.1016/j.molcel.2019.07.028. [7] RAMACHANDRAN P, DOBIE R, WILSON-KANAMORI JR, et al. Resolving the fibrotic niche of human liver cirrhosis at single-cell level[J]. Nature, 2019, 575( 7783): 512- 518. DOI: 10.1038/s41586-019-1631-3. [8] MAGEE N, AHAMED F, EPPLER N, et al. Hepatic transcriptome profiling reveals early signatures associated with disease transition from non-alcoholic steatosis to steatohepatitis[J]. Liver Res, 2022, 6( 4): 238- 250. DOI: 10.1016/J.LIVRES.2022.11.001. [9] YANG B, LUO W, WANG MX, et al. Macrophage-specific MyD88 deletion and pharmacological inhibition prevents liver damage in non-alcoholic fatty liver disease via reducing inflammatory response[J]. Biochim Biophys Acta Mol Basis Dis, 2022, 1868( 10): 166480. DOI: 10.1016/j.bbadis.2022.166480. [10] BINATTI E, GERUSSI A, BARISANI D, et al. The role of macrophages in liver fibrosis: New therapeutic opportunities[J]. Int J Mol Sci, 2022, 23( 12): 6649. DOI: 10.3390/ijms23126649. [11] XU L, CHEN YP, NAGASHIMADA M, et al. CC chemokine ligand 3 deficiency ameliorates diet-induced steatohepatitis by regulating liver macrophage recruitment and M1/M2 status in mice[J]. Metabolism, 2021, 125: 154914. DOI: 10.1016/j.metabol.2021.154914. [12] XIANG P. Zhx2 promotes NAFLD by enhancing pro-inflammatory responses of macrophag[D]. Jinan: Shandong University, 2021. DOI: 10.27272/d.cnki.gshdu.2021.005369.向鹏. Zhx2促进巨噬细胞炎症反应参与NAFLD的作用及机制研究[D]. 济南: 山东大学, 2021. DOI: 10.27272/d.cnki.gshdu.2021.005369. [13] BANG BR, MIKI H, KANG YJ. Mitochondrial PGAM5-Drp1 signaling regulates the metabolic reprogramming of macrophages and regulates the induction of inflammatory responses[J]. Front Immunol, 2023, 14: 1243548. DOI: 10.3389/fimmu.2023.1243548. [14] XI ZQ, BAO MW. Research progress on glycolipid metabolism reprogramming of macrophage in non-alcoholic fatty liver disease[J]. Curr Biotechnol, 2024, 14( 3): 399- 405. DOI: 10.19586/j.2095-2341.2023.0164.席照青, 包明威. 巨噬细胞糖脂代谢重编程在非酒精性脂肪肝中的研究进展[J]. 生物技术进展, 2024, 14( 3): 399- 405. DOI: 10.19586/j.2095-2341.2023.0164. [15] INOMATA Y, OH JW, TANIGUCHI K, et al. Downregulation of miR-122-5p activates glycolysis via PKM2 in kupffer cells of rat and mouse models of non-alcoholic steatohepatitis[J]. Int J Mol Sci, 2022, 23( 9): 5230. DOI: 10.3390/ijms23095230. [16] LI JY, CHEN XC, SONG SY, et al. Hexokinase 2-mediated metabolic stress and inflammation burden of liver macrophages via histone lactylation in MASLD[J]. Cell Rep, 2025, 44( 3): 115350. DOI: 10.1016/j.celrep.2025.115350. [17] ZIZZO G, COHEN PL. The PPAR-γ antagonist GW9662 elicits differentiation of M2c-like cells and upregulation of the MerTK/Gas6 axis: A key role for PPAR-γ in human macrophage polarization[J]. J Inflamm, 2015, 12: 36. DOI: 10.1186/s12950-015-0081-4. [18] CHEN YX, WU YP, ZHANG Y, et al. Modulation of lipid metabolism and Keap1-Nrf2 pathway activation in macrophages by targeting PPARγ affects NAFLD progression[J]. J Gastroenterol Hepatol, 2025, 40( 8): 2119- 2133. DOI: 10.1111/jgh.17033. [19] WU YL, LIN ZJ, LI CC, et al. Epigenetic regulation in metabolic diseases: Mechanisms and advances in clinical study[J]. Signal Transduct Target Ther, 2023, 8( 1): 98. DOI: 10.1038/s41392-023-01333-7. [20] YU PJ, XIAN YX, CAO HY, et al. Lipotoxicity-induced TET1 mediates CD36 DNA demethylation in liver macrophages to aggravate NAFLD progression[J]. Diabetes, 2024, 73( Supplement_1): 1572-P. DOI: 10.2337/db24-1572-p. [21] LEE J, AN H, KIM CS, et al. The methyltransferase MLL4 promotes nonalcoholic steatohepatitis by enhancing NF-κB signaling[J]. J Biol Chem, 2024, 300( 12): 107984. DOI: 10.1016/j.jbc.2024.107984. [22] PENTAGNA N, PINHEIRO DA COSTA T, SOARES DOS SANTOS CARDOSO F, et al. Epigenetic control of myeloid cells behavior by Histone Deacetylase activity(HDAC) during tissue and organ regeneration in Xenopus laevis[J]. Dev Comp Immunol, 2021, 114: 103840. DOI: 10.1016/j.dci.2020.103840. [23] PAN Q, XUE R, FAN JG. Macrographic phenotype of docosahexaenoic acid metabolism plays a role in nonalcoholic steatohepatitis[J]. J Pract Hepatol, 2024, 27( 1): 155- 158. DOI: 10.3969/j.issn.1672-5069.2024.01.040.潘勤, 薛芮, 范建高. 巨噬细胞DHA代谢表型在非酒精性脂肪性肝炎发病过程中的作用[J]. 实用肝脏病杂志, 2024, 27( 1): 155- 158. DOI: 10.3969/j.issn.1672-5069.2024.01.040. [24] KIMBROUGH D, WANG SH, WRIGHT LH, et al. HDAC inhibition helps post-MI healing by modulating macrophage polarization[J]. J Mol Cell Cardiol, 2018, 119: 51- 63. DOI: 10.1016/j.yjmcc.2018.04.011. [25] WANG Y, LI X, CHEN Q, et al. Histone deacetylase 6 regulates the activation of M1 macrophages by the glycolytic pathway during acute liver failure[J]. J Inflamm Res, 2021, 14: 1473- 1485. DOI: 10.2147/JIR.S302391. [26] PARTHASARATHY G, REVELO X, MALHI H. Pathogenesis of nonalcoholic steatohepatitis: An overview[J]. Hepatol Commun, 2020, 4( 4): 478- 492. DOI: 10.1002/hep4.1479. [27] LI L, CUI L, LIN P, et al. Kupffer-cell-derived IL-6 is repurposed for hepatocyte dedifferentiation via activating progenitor genes from injury-specific enhancers[J]. Cell Stem Cell, 2023, 30( 3): 283- 299. e 9. DOI: 10.1016/j.stem.2023.01.009. [28] LI YQ, TANG WJ, ZHOU YJ. Role of intestinal microbiota and metabolites in the development, progression, and treatment of nonalcoholic fatty liver disease[J]. J Clin Hepatol, 2023, 39( 8): 1805- 1810. DOI: 10.3969/j.issn.1001-5256.2023.08.006.李永强, 唐文娟, 周永健. 肠道菌群及其代谢产物在非酒精性脂肪性肝病发生发展及治疗中的作用[J]. 临床肝胆病杂志, 2023, 39( 8): 1805- 1810. DOI: 10.3969/j.issn.1001-5256.2023.08.006. [29] SHARMA S, TIWARI N, TANWAR SS. The current findings on the gut-liver axis and the molecular basis of NAFLD/NASH associated with gut microbiome dysbiosis[J]. Naunyn Schmiedebergs Arch Pharmacol, 2025, 398( 9): 11541- 11579. DOI: 10.1007/s00210-025-04069-z. [30] NAGASHIMADA M, HONDA M. Effect of microbiome on non-alcoholic fatty liver disease and the role of probiotics, prebiotics, and biogenics[J]. Int J Mol Sci, 2021, 22( 15): 8008. DOI: 10.3390/ijms22158008. [31] MA LQ, LI HG, HU JB, et al. Indole alleviates diet-induced hepatic steatosis and inflammation in a manner involving myeloid cell 6-phosphofructo-2-kinase/fructose-2, 6-biphosphatase 3[J]. Hepatology, 2020, 72( 4): 1191- 1203. DOI: 10.1002/hep.31115. [32] SHAO JW, GE TT, WEI YL, et al. Co-interventions with Clostridium butyricum and soluble dietary fiber targeting the gut microbiota improve MAFLD via the Acly/Nrf2/NF-κB signaling pathway[J]. Food Funct, 2022, 13( 10): 5807- 5819. DOI: 10.1039/d1fo04224f. [33] HUANG XY, YAO YC, HOU XL, et al. Macrophage SCAP contributes to metaflammation and lean NAFLD by activating STING-NF-κB signaling pathway[J]. Cell Mol Gastroenterol Hepatol, 2022, 14( 1): 1- 26. DOI: 10.1016/j.jcmgh.2022.03.006. [34] RUSSO-SAVAGE L, SCHULMAN IG. Liver X receptors and liver physiology[J]. Biochim Biophys Acta Mol Basis Dis, 2021, 1867( 6): 166121. DOI: 10.1016/j.bbadis.2021.166121. [35] KAZANKOV K, JØRGENSEN SMD, THOMSEN KL, et al. The role of macrophages in nonalcoholic fatty liver disease and nonalcoholic steatohepatitis[J]. Nat Rev Gastroenterol Hepatol, 2019, 16( 3): 145- 159. DOI: 10.1038/s41575-018-0082-x. [36] DAEMEN S, GAINULLINA A, KALUGOTLA G, et al. Dynamic shifts in the composition of resident and recruited macrophages influence tissue remodeling in NASH[J]. Cell Rep, 2021, 34( 2): 108626. DOI: 10.1016/j.celrep.2020.108626. [37] AKKıZ H, GIESELER RK, CANBAY A. Liver fibrosis: From basic science towards clinical progress, focusing on the central role of hepatic stellate cells[J]. Int J Mol Sci, 2024, 25( 14): 7873. DOI: 10.3390/ijms25147873. [38] WEI HR, XIAO F, WEI HS. Research progress on the mechanism of endoplasmic reticulum stress in metabolic dysfunction-associated fatty liver disease[J/CD]. Chin J Liver Dis: Electronic Edition, 2024, 16( 1): 13- 17. DOI: 10.3969/j.issn.1674-7380.2024.01.003.韦何锐, 肖凡, 魏红山. 内质网应激在代谢相关脂肪性肝病中的作用机制研究进展[J/CD]. 中国肝脏病杂志(电子版), 2024, 16( 1): 13- 17. DOI: 10.3969/j.issn.1674-7380.2024.01.003. [39] GUO C, ZHENG LL, WANG L, et al. Application prospect for extracellular vesicles in nonalcoholic steatohepatitis[J]. Chem Life, 2023, 43( 11): 1699- 1706. DOI: 10.13488/j.smhx.20230336.郭才, 郑兰兰, 王莉, 等. 细胞外囊泡在非酒精性脂肪性肝炎中的应用前景[J]. 生命的化学, 2023, 43( 11): 1699- 1706. DOI: 10.13488/j.smhx.20230336. [40] FIZANNE L, VILLARD A, BENABBOU N, et al. Faeces-derived extracellular vesicles participate in the onset of barrier dysfunction leading to liver diseases[J]. J Extracell Vesicles, 2023, 12( 2): 12303. DOI: 10.1002/jev2.12303. [41] GUILLIAMS M, SCOTT CL. Liver macrophages in health and disease[J]. Immunity, 2022, 55( 9): 1515- 1529. DOI: 10.1016/j.immuni.2022.08.002. [42] PUENGEL T, LEFERE S, HUNDERTMARK J, et al. Combined therapy with a CCR2/CCR5 antagonist and FGF21 analogue synergizes in ameliorating steatohepatitis and fibrosis[J]. Int J Mol Sci, 2022, 23( 12): 6696. DOI: 10.3390/ijms23126696. [43] DONG XY, FENG YT, XU DQ, et al. Targeting macrophagic 17β-HSD7 by fenretinide for the treatment of nonalcoholic fatty liver disease[J]. Acta Pharm Sin B, 2023, 13( 1): 142- 156. DOI: 10.1016/j.apsb.2022.04.003. [44] ZHANG MY, LI K, HUANG XX, et al. Macrophage Notch1 signaling modulates regulatory T cells via the TGFB axis in early MASLD[J]. JHEP Rep, 2025, 7( 1): 101242. DOI: 10.1016/j.jhepr.2024.101242. [45] ROH YS, KIM JW, PARK S, et al. Toll-like receptor-7 signaling promotes nonalcoholic steatohepatitis by inhibiting regulatory T cells in mice[J]. Am J Pathol, 2018, 188( 11): 2574- 2588. DOI: 10.1016/j.ajpath.2018.07.011. [46] WANG H, ZHANG HJ, WANG Y, et al. Regulatory T-cell and neutrophil extracellular trap interaction contributes to carcinogenesis in non-alcoholic steatohepatitis[J]. J Hepatol, 2021, 75( 6): 1271- 1283. DOI: 10.1016/j.jhep.2021.07.032. [47] HAN JM, PATTERSON SJ, SPECK M, et al. Insulin inhibits IL-10-mediated regulatory T cell function: Implications for obesity[J]. J Immunol, 2014, 192( 2): 623- 629. DOI: 10.4049/jimmunol.1302181. [48] WANG B, LI XP, HU WJ, et al. Silencing of lncRNA SNHG20 delays the progression of nonalcoholic fatty liver disease to hepatocellular carcinoma via regulating liver Kupffer cells polarization[J]. IUBMB Life, 2019, 71( 12): 1952- 1961. DOI: 10.1002/iub.2137. [49] CHEN T, CHEN Y. Research progress in mechanism of fibrosis regulated by macrophage polarization[J]. J Jilin Univ: Med Edit, 2024, 50( 5): 1465- 1473. DOI: 10.13481/j.1671-587X.20240534.陈潭, 陈艳. 巨噬细胞极化调控纤维化机制的研究进展[J]. 吉林大学学报(医学版), 2024, 50( 5): 1465- 1473. DOI: 10.13481/j.1671-587X.20240534. [50] WAN ZP, YANG XA, LIU XQ, et al. M2 macrophage-derived exosomal microRNA-411-5p impedes the activation of hepatic stellate cells by targeting CAMSAP1 in NASH model[J]. iScience, 2022, 25( 7): 104597. DOI: 10.1016/j.isci.2022.104597. [51] CAO Y, MAI WL, LI R, et al. Macrophages evoke autophagy of hepatic stellate cells to promote liver fibrosis in NAFLD mice via the PGE2/EP4 pathway[J]. Cell Mol Life Sci, 2022, 79( 6): 303. DOI: 10.1007/s00018-022-04319-w. [52] ARELAKI S, KOLETSA T, SINAKOS E, et al. Neutrophil extracellular traps enriched with IL-1β and IL-17A participate in the hepatic inflammatory process of patients with non-alcoholic steatohepatitis[J]. Virchows Arch, 2022, 481( 3): 455- 465. DOI: 10.1007/s00428-022-03330-7. [53] JORCH SK, KUBES P. An emerging role for neutrophil extracellular traps in noninfectious disease[J]. Nat Med, 2017, 23( 3): 279- 287. DOI: 10.1038/nm.4294. [54] HONDA M, KUBES P. Neutrophils and neutrophil extracellular traps in the liver and gastrointestinal system[J]. Nat Rev Gastroenterol Hepatol, 2018, 15( 4): 206- 221. DOI: 10.1038/nrgastro.2017.183. [55] MARETTI-MIRA AC, GOLDEN-MASON L, SALOMON MP, et al. Cholesterol-induced M4-like macrophages recruit neutrophils and induce NETosis[J]. Front Immunol, 2021, 12: 671073. DOI: 10.3389/fimmu.2021.671073. [56] PARK J, LANGMEAD CJ, RIDDY DM. New advances in targeting the resolution of inflammation: Implications for specialized pro-resolving mediator GPCR drug discovery[J]. ACS Pharmacol Transl Sci, 2020, 3( 1): 88- 106. DOI: 10.1021/acsptsci.9b00075. [57] TREINER E, DUBAN L, BAHRAM S, et al. Selection of evolutionarily conserved mucosal-associated invariant T cells by MR1[J]. Nature, 2003, 422( 6928): 164- 169. DOI: 10.1038/nature01433. [58] LI YM, HUANG BY, JIANG X, et al. Mucosal-associated invariant T cells improve nonalcoholic fatty liver disease through regulating macrophage polarization[J]. Front Immunol, 2018, 9: 1994. DOI: 10.3389/fimmu.2018.01994. -



 PDF下载 ( 1646 KB)
PDF下载 ( 1646 KB)

 下载:
下载: