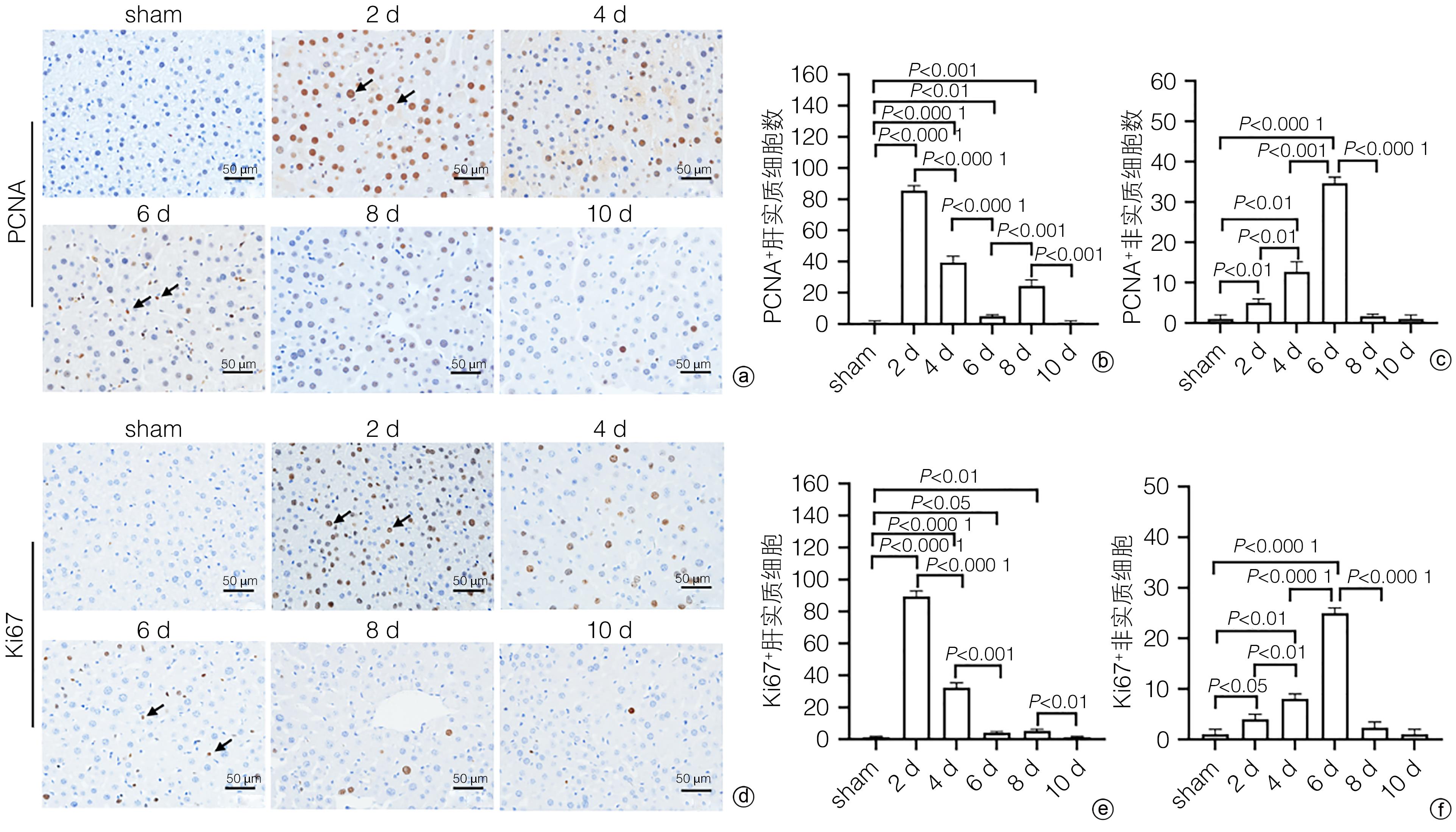
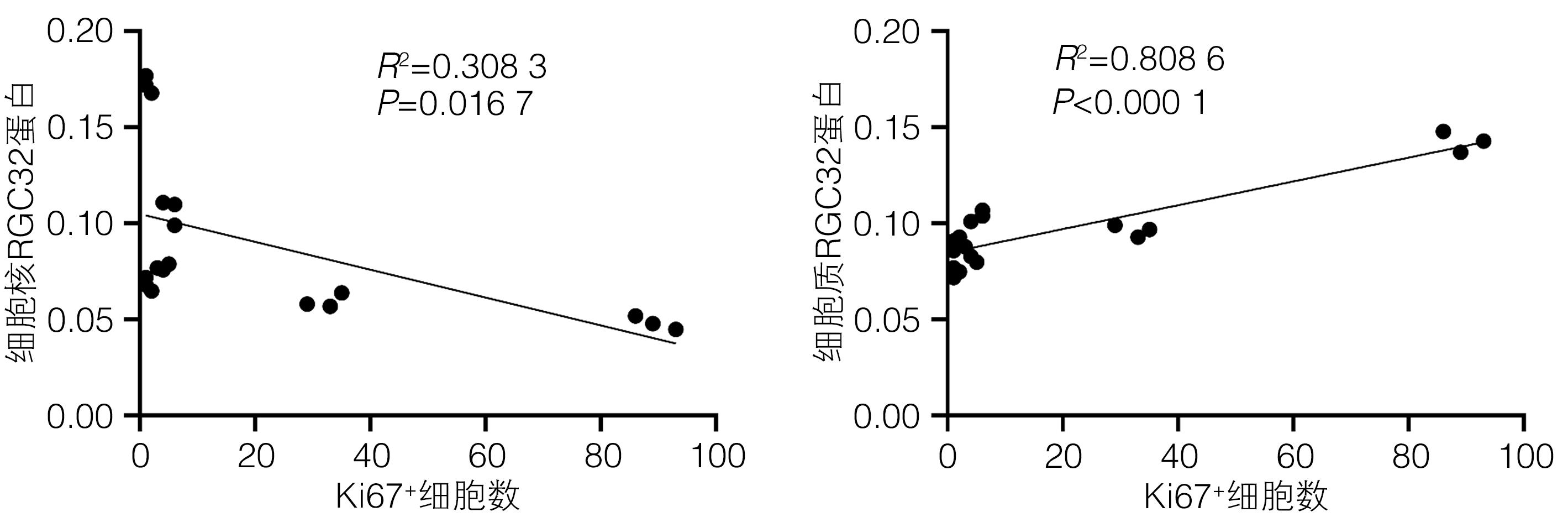
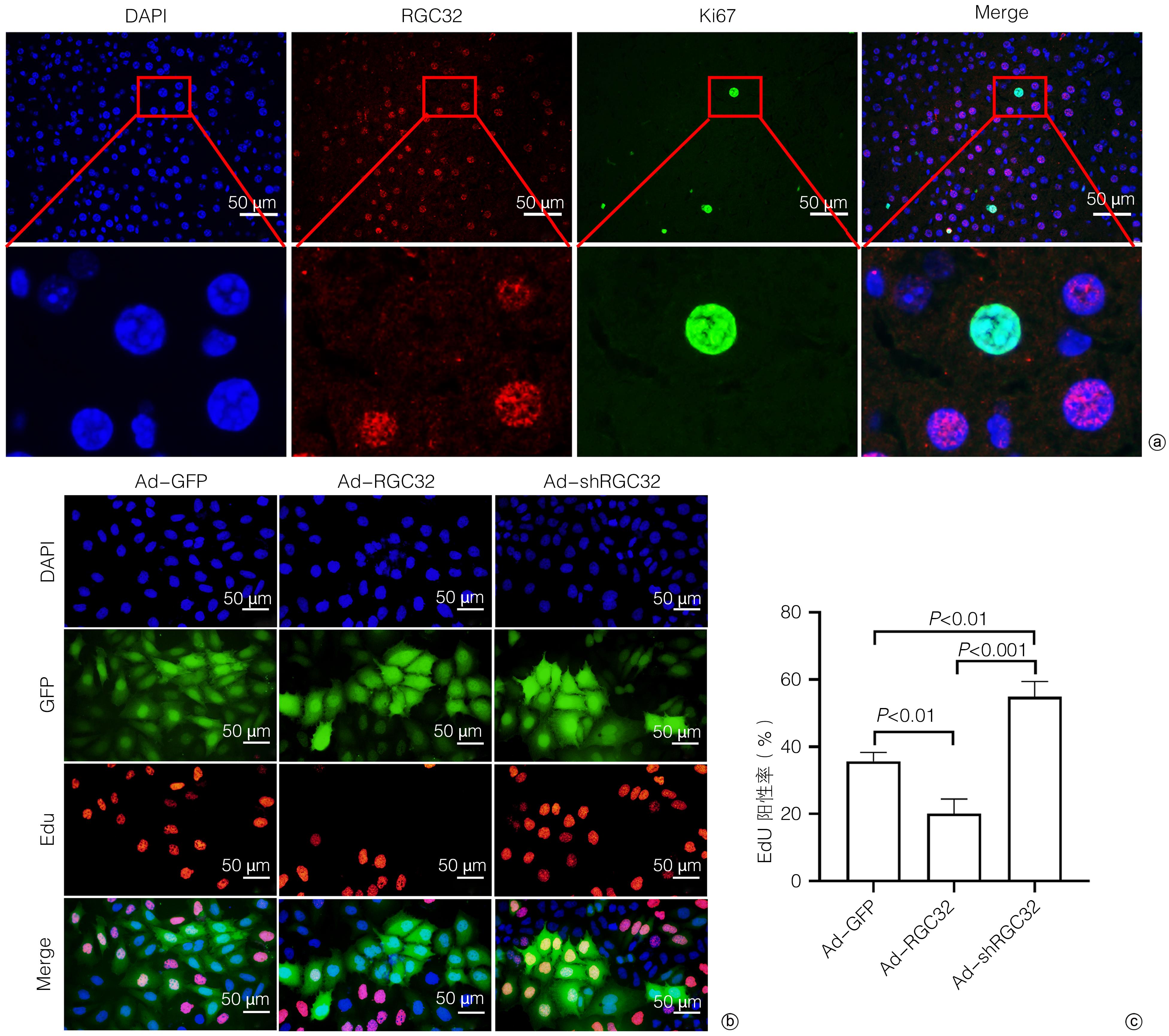
| [1] |
MICHALOPOULOS GK, BHUSHAN B. Liver regeneration: biological and pathological mechanisms and implications[J]. Nat Rev Gastroenterol Hepatol, 2021, 18( 1): 40- 55. DOI: 10.1038/s41575-020-0342-4. |
| [2] |
LIN S, NASCIMENTO EM, GAJERA CR, et al. Distributed hepatocytes expressing telomerase repopulate the liver in homeostasis and injury[J]. Nature, 2018, 556( 7700): 244- 248. DOI: 10.1038/s41586-018-0004-7. |
| [3] |
CHEN HX, HUANG SJ, CHEN JY, et al. Role of complement in liver injury and regeneration[J]. Central South Pharm, 2021, 19( 10): 2123- 2127. DOI: 10.7539/j.issn.1672-2981.2021.10.023. |
| [4] |
WANG Q, QU X. New insights into the roles of RGC32[J]. Cell Mol Immunol, 2018, 15( 8): 803- 804. DOI: 10.1038/cmi.2017.154. |
| [5] |
|
| [6] |
NEVZOROVA YA, TOLBA R, TRAUTWEIN C, et al. Partial hepatectomy in mice[J]. Lab Anim, 2015, 49( Suppl 1): 81- 88. DOI: 10.1177/0023677215572000. |
| [7] |
LÓPEZ-LUQUE J, FABREGAT I. Revisiting the liver: from development to regeneration-what we ought to know![J]. Int J Dev Biol, 2018, 62( 6- 7- 8): 441- 451. DOI: 10.1387/ijdb.170264JL. |
| [8] |
van HAELE M, SNOECK J, ROSKAMS T. Human liver regeneration: an etiology dependent process[J]. Int J Mol Sci, 2019, 20( 9): 2332. DOI: 10.3390/ijms20092332. |
| [9] |
YAGI S, HIRATA M, MIYACHI Y, et al. Liver regeneration after hepatectomy and partial liver transplantation[J]. Int J Mol Sci, 2020, 21( 21): 8414. DOI: 10.3390/ijms21218414. |
| [10] |
YU ZY, LIN X, HAN Y, et al. Role of liver sinusoidal endothelial cells in liver regeneration and the development of liver fibrosis[J]. J Clin Hepatol, 2019, 35( 9): 2072- 2074. DOI: 10.3969/j.issn.1001-5256.2019.09.041. |
| [11] |
LI N, LIU C, MA G, et al. Asparaginyl endopeptidase may promote liver sinusoidal endothelial cell angiogenesis via PI3K/Akt pathway[J]. Rev Esp Enferm Dig, 2019, 111( 3): 214- 222. DOI: 10.17235/reed.2018.5709/2018. |
| [12] |
VLAICU SI, TATOMIR A, ANSELMO F, et al. RGC32 and diseases: the first 20 years[J]. Immunol Res, 2019, 67( 2- 3): 267- 279. DOI: 10.1007/s12026-019-09080-0. |
| [13] |
|
| [14] |
VLAICU SI, TATOMIR A, BOODHOO D, et al. RGC32 is expressed in the human atherosclerotic arterial wall: Role in C5b-9-induced cell proliferation and migration[J]. Exp Mol Pathol, 2016, 101( 2): 221- 230. DOI: 10.1016/j.yexmp.2016.09.004. |
| [15] |
CUI XB, GUO X, CHEN SY. Response gene to complement 32 deficiency causes impaired placental angiogenesis in mice[J]. Cardiovasc Res, 2013, 99( 4): 632- 639. DOI: 10.1093/cvr/cvt121. |
| [16] |
CUI XB, LUAN JN, DONG K, et al. RGC32(response gene to complement 32) deficiency protects endothelial cells from inflammation and attenuates atherosclerosis[J]. Arterioscler Thromb Vasc Biol, 2018, 38( 4): e36-e47. DOI: 10.1161/ATVBAHA.117.310656. |
| [17] |
HU C, WU Z, LI L. Mesenchymal stromal cells promote liver regeneration through regulation of immune cells[J]. Int J Biol Sci, 2020, 16( 5): 893- 903. DOI: 10.7150/ijbs.39725. |
| [18] |
ZHENG J, LI JT, JIN J, et al. Characteristics of incision bacterial infection and changes of immune cytokines in patients with hepatocellular carcinoma after hepatectomy[J]. Chin J Microecol, 2022, 34( 7): 819- 823, 845. DOI: 10.13381/j.cnki.cjm.202207014. |
| [19] |
SAIGUSA K, IMOTO I, TANIKAWA C, et al. RGC32, a novel p53-inducible gene, is located on centrosomes during mitosis and results in G2/M arrest[J]. Oncogene, 2007, 26( 8): 1110- 1121. DOI: 10.1038/sj.onc.1210148. |



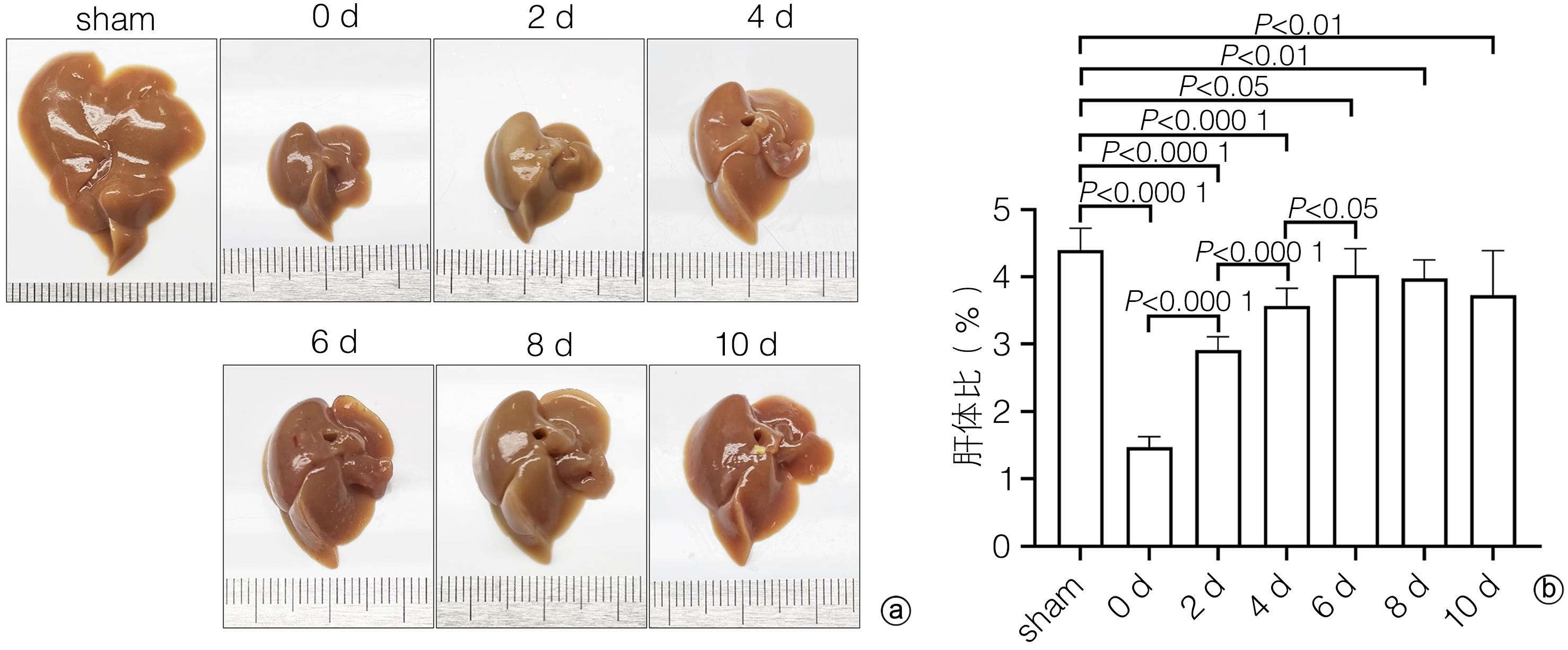




 DownLoad:
DownLoad: